Current PhD vacancies
Apply now!
International PhD Call for Fully Funded PhD Studentships within our 18 Doctoral Programmes
Our university offers PhD research opportunities in the fields of Life Sciences, Medical Engineering and AI/Machine Learning, in the current PhD call. You can explore currently available positions here on this website. To ensure a fair and transparent recruitment process where all candidates have equal opportunities, we can only consider applications submitted through our online application tool for the listed projects. It is NOT necessairy to reach out in advance to any PI participating in the PhD call. If you have any questions about the projects or process, please reach out to phdrecruitment@meduniwien.ac.at.
Current PhD Projects Available
AI, Imaging & Biomedical Engineering
- AI models using imaging and genetics to predict breast cancer risk and tumour aggressiveness in BRCA variant carriers
- Digital biomarkers for enhanced AI-guided therapy in heart failure (D-BEAT)
- Experimental models for optimizing cardiovascular implant surfaces towards enhanced hemocompatibility
Inflammation, Immunology & Cardiovascular Disease
- Thrombo-inflammatory drug candidates in humanized mouse models of abdominal aortic aneurysms
- Bacterial extracellular vesicles as drivers of haemostatic complications in infection?
- From infection to thrombosis: mechanisms linking infection and coagulation
- Impact of immunothrombosis on pulmonary disease manifestations
- Platelets as biomarkers for safer blood-contacting medical devices
- Effect of endothelial interaction with circulating blood cells in thrombus resolution and post-thrombotic disorders
- Elucidating the role of IgG antibodies in atherosclerosis
- The role of T cells in atherosclerosis and heart attacks
- Clonal hematopoiesis in chronic inflammation and cardiovascular disease
- Linking inflammatory and metabolic changes to procoagulant phenotypes and clinical outcomes in thrombosis
- Impact of mechanical stress on macrophages in atherosclerotic lesions
- Towards development of fibroblast-targeting therapeutic approaches in systemic sclerosis (SSc)
- Ashwagandha Supplementation in Endometriosis: A Randomized Controlled Trial Integrating Clinical and Mechanistic Outcomes
- Clinical and Translational Research in Inherited Bleeding Disorders, with Emphasis on Hemophilia
- Role of histamine in the pathophysiology of thrombosis in histamine sensitive species
Metabolism, Kidney & Systemic Disease
- Metaflammation in Kidney Diseases and Dialysis
Molecular Biology, Transport & Cancer Mechanisms
- Molecular understanding of chemotherapy response and resistance in colorectal cancer using complex organoids
- Studying the molecular mechanism of the lysosomal dipeptide transporter MFSD1
- Biophysical properties of organic cation transporter 3 (OCT3)
- Hormone replacement therapy and breast cancer risk and outcomes in germline BRCA1 and BRCA2 variant carriers
Neuroscience & Systems Biology
- Prefrontal circuits for value-based decision making
- The role of neuromodulation in prosocial behavior in health and disease
- Validation of novel peripheral pain targets
- The impact of dyslipidemia on hematoiesis in the skull
- MRI-based in vivo histology of Multiple Sclerosis
General Requirements
To apply, you must hold a Master’s or MD degree (or equivalent) in a field relevant to the open PhD project(s). You may apply while completing your Master’s, provided you expect to finish within six months.
Other essential qualifications include:
- Strong academic performance and relevant research experience
- Proficiency in English (written and spoken)
- Experience in writing and presenting scientific results, with clear data interpretation
- Motivation to work in an interdisciplinary, collaborative international research environment
- Motivation to develop and carry out your own research project
What Makes a Strong Application
You are particularly well-suited if you also have:
- A Master’s or MD degree completed within the past three years
- Research experience directly related to the project(s), for example through a thesis or research project
- Practical experience—experimental and/or computational—relevant to the topic
- Evidence of prior research output, such as reports, theses, or publications
We welcome applications from candidates with diverse backgrounds and career paths. If your experience does not match every point exactly but you can demonstrate a strong and relevant connection to the project, we encourage you to apply.
Start Your Application Early
We recommend starting your application well in advance of the deadline. Preparing your materials and securing referee submissions can take time, particularly as referees must complete their evaluations directly in the system. Starting early helps ensure your application is complete and submitted on time. If you are unsure who can act as a referee, please consult the FAQ.
We are looking forward to receive your application!
We will add new PhD positions till 30.03.2026 and encourage all eligible candidates to apply by the specified deadline (26.04.2026) to ensure equal consideration. Application guideline for the online tool can be looked up here.
- AI/ML
- Bioinformatics
- Cancer
- Computer lab
- Data Science
HER-CARE is a EU funded doctoral network developing novel approaches to support care for breast cancer. In this PhD project you will develop AI models using imaging together with multi-modal data (e.g., genetics) to predict individual breast cancer progression and tumour aggressiveness. A specific focus is on the linking of modalities from clinical-, genetic-, and multi-parametric MRI data for prediction and the investigation of relationships across modalities. Relevant methods range from vision language models (VLM), multi agent systems (MAS), to transformer models for the representation and structure learning in complex multi-modal data. For instance, we are curious if MAS can be used for biological mechanism discovery from patient data, or if hyper-models can steer the prediction from multi-modal but incomplete patient data.
The PhD project will be embedded in a European doctoral network and a local environment with strong expertise in ML (CIR Lab https://www.cir.meduniwien.ac.at , and the Comprehensive Center for AI in Medicine, with more than 25 AI labs https://caim.meduniwien.ac.at), imaging, and breast cancer research. It will include stays with industry and academic partners in the project. You will be embedded in an interdisciplinary research team with members from machine learning, biomedical technology, medical imaging, and cancer medicine. Your interactions will include close ties to clinicians for feedback, collaboration, and the development of ideas. Students from these fields will be in the same lab. Cutting-edge computing environment including multiple DGX systems will be available.
Skills to be acquired during PhD training
During the PhD project, the student will gain interdisciplinary training in artificial intelligence, medical imaging analysis, and multi-modal biomedical data integration, including:
- Development of machine learning and deep learning models for medical imaging and biomedical data
- Design and implementation of transformer-based architectures, vision–language models, and multi-agent systems
- Integration and analysis of multi-modal biomedical data, including imaging, clinical, and genetic datasets
- Application of multi-parametric MRI analysis for breast cancer research
- Development of predictive models for tumour progression and aggressiveness
- Representation learning and modelling of complex and incomplete biomedical datasets
- Collaboration with clinicians to translate machine learning approaches into clinical research contexts
- Development of scientific communication and interdisciplinary collaboration skills in AI and biomedical research
PhD network and research environment
The project is part of HER-CARE, a European doctoral network funded by the European Union that focuses on developing innovative data-driven approaches to improve breast cancer care.
The PhD student will be embedded in an interdisciplinary research environment at the Medical University of Vienna and benefit from:
- Collaboration with the Computational Imaging Research Lab (CIR Lab) and the Comprehensive Center for AI in Medicine, which includes more than 25 AI research laboratories
- Access to a cutting-edge computing infrastructure, including DGX systems for large-scale machine learning
- Close collaboration with experts in machine learning, biomedical engineering, medical imaging, and oncology
- Direct interaction with clinicians for feedback and clinical relevance of developed models
- Participation in an international doctoral training network, including secondments with academic and industry partners
- A vibrant interdisciplinary research environment combining AI, imaging, and cancer research
Master’s degree in relevant fields such as Computer Science, Biomedical Engineering, Bioinformatics, Data Science, or a related discipline.
Experience in Machine Learning techniques, including deep learning, and a strong interest in medical applications, (breast) cancer biology, medical imaging such as MRI are a plus.
Motivation to develop novel machine learning approaches in the field of medical imaging and precision medicine. Interest in interdisciplinary work at the interface of AI and medicine.
Applicants must meet the MSCA Doctoral Network eligibility criteria:
- A Master’s degree
- No PhD degree at the time of recruitment
- Mobility rule: candidates must not have lived, worked, or studied in the country of the recruiting beneficiary for more than 12 months in the 36 months prior to recruitment. Any nationality is eligible, provided the mobility rule is fulfilled
- Human research
- Immunology
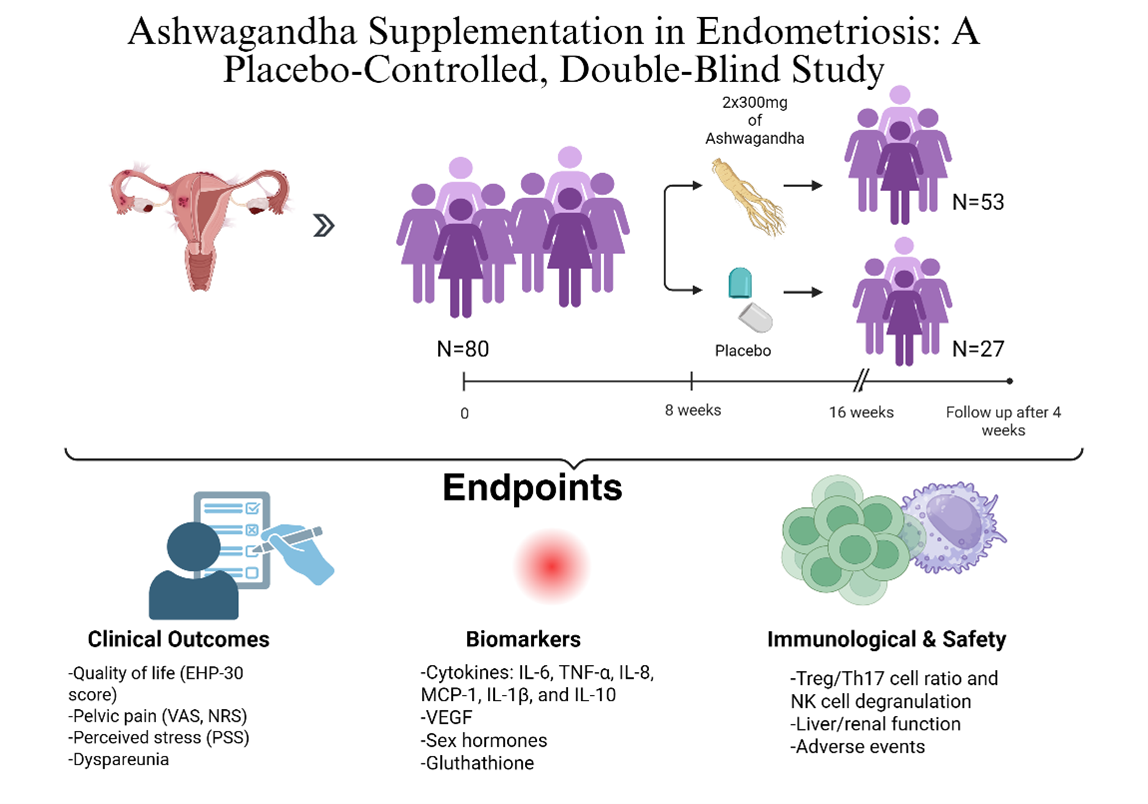
This PhD project investigates the therapeutic potential of Ashwagandha supplementation in women with endometriosis within a randomized, double-blind, placebo-controlled clinical trial. Endometriosis is a chronic, inflammatory condition associated with persistent pelvic pain, impaired quality of life, and limited long-term treatment options. Increasing evidence suggests that plant-derived compounds with anti-inflammatory and immunomodulatory properties may represent promising complementary strategies in the management of this disease.
The project integrates patient-oriented clinical research with applied translational laboratory approaches. The PhD candidate will be actively involved in patient recruitment, interaction, and clinical data collection within a structured clinical trial setting. In parallel, the candidate will perform laboratory analyses on human biological samples, including immune cell phenotyping and biomarker assessment, enabling the investigation of underlying biological mechanisms.
This interdisciplinary framework provides a unique opportunity to directly link clinical outcomes with mechanistic insights. The candidate will gain hands-on experience in the conduct of clinical trials, data analysis, and modern laboratory techniques in immunology and vascular biology, while developing a strong translational research perspective.
Skills to be acquired during PhD training
During this PhD project, the candidate will gain comprehensive, hands-on training at the interface of clinical research and applied biomedical science. The structured combination of patient-oriented work and laboratory-based analyses offers a unique opportunity to develop a broad and highly relevant skill set. The candidate will be actively involved in the full conduct of a randomized, double-blind, placebo-controlled clinical trial, including patient recruitment, informed consent procedures, and high-quality clinical documentation. This provides direct exposure to real-world clinical research workflows and regulatory standards.
In parallel, the candidate will acquire practical laboratory experience through the analysis of human biological samples. This includes cytokine profiling using techniques such as multiplex assays and ELISA, as well as immune phenotyping approaches to characterize relevant cellular responses. These methods will allow the candidate to explore biological mechanisms underlying clinical outcomes.
A strong emphasis is placed on data handling, analysis, and interpretation. The candidate will learn how to integrate clinical endpoints with laboratory-derived data, enabling a deeper understanding of disease mechanisms and therapeutic effects. In addition, the candidate will have the opportunity to contribute to other ongoing projects within the department, allowing them to broaden their expertise across different research areas and methodologies. This exposure will further strengthen their scientific versatility and interdisciplinary perspective.
Importantly, the project fosters the development of a translational research mindset, teaching the candidate how to connect patient-centered observations with mechanistic insights. This combination of clinical and analytical expertise represents a highly valuable skill set for future careers in academia, clinical research, or the pharmaceutical and biotechnology sectors.
PhD network and research environment
The project is embedded within the Department of Clinical Pharmacology at the Medical University of Vienna, offering access to well-established clinical research infrastructure, state-of-the-art core facilities, and a highly collaborative environment. Close cooperation with the Department of General Gynaecology and Gynaecological Oncology ensures direct clinical relevance and integration into ongoing research activities.
• Degree in Medicine (completed medical studies). No other degrees can be taken into consideration for this project
• Proficiency in German and English
• Interest in laboratory work
- Animal research
- Cardiovascular systems
- Human research
- Immunology
- Molecular and cell biology
- Wet lab
Haemostasis and immunity are closely interconnected systems with platelets representing an essential link as they not only fulfil crucial functions in haemostasis and thrombosis, but also rapidly respond to microbial components and drive inflammatory host responses. Hence, haemostatic disturbances such as thrombocytopenia, microthrombosis or bleeding events are frequently observed during infections and associated with disease severity and outcome.
Our group investigates the molecular crosstalk between platelets, leukocytes and pathogens, aiming to better understand the consequences of these interactions for the pro-thrombotic and immunomodulatory roles of platelets in inflammation and infection.
Bacteria can modulate physiologic host responses by shedding bacterial extracellular vesicles (bEVs) which serve as bacterial vanguards, rapidly distributing throughout the tissue and disarming host defences before invasion of parent bacteria. In previous experiments we found that bEVs of Vibrio cholerae and various other Vibrio species critically impaired central platelet functions, thereby demonstrating that bEVs may also impact on primary haemostasis.
In the proposed PhD project, we aim to elucidate the molecular mechanisms of V. cholerae bEV-mediated platelet inhibition and unravel how these bEVs contribute to haemostatic and immunothrombotic complications in bacterial infection. The successful PhD candidate will combine state-of-the-art in vitro assays with cutting-edge imaging techniques and advanced in vivo models to investigate the molecular mechanisms underlying bEV-platelet interactions and their effects on haemostasis and host immunity.
Skills to be acquired during PhD training
During the PhD project, the student will acquire extensive training in a variety of experimental techniques and soft skills, including:
- Isolation and characterization of bacterial extracellular vesicles as well as human and murine primary cells (platelets, leukocytes)
- State-of-the art wet-lab techniques for studying platelet and leukocyte activation status and aberrations of intracellular signalling processes, such as multi-colour flow cytometry, (super-resolution) fluorescence microscopy, isolation of cytoskeletal compartments and standard molecular biology techniques (e.g. ELISA, Western Blot, …)
- High-throughput analysis of intracellular signalling events via proteomics and phospho-proteomics
- Specialized platelet function analyses (e.g. light transmission aggregometry, live cell microscopy for in vitro thrombosis assays under flow)
- Murine in vivo models of thrombosis, bleeding and infection
- Scientific writing and presentation of research data in an international and interdisciplinary research environment (in-house seminars, conferences)
- Critical evaluation of scientific data and literature
PhD network and research environment
The PhD project will be conducted at the Centre for Physiology and Pharmacology, one of the largest preclinical organisational units of the Medical University of Vienna (MUV). The research group is further embedded within the university’s Cardiovascular Medicine Research Cluster, providing an international and multi-disciplinary environment with regular networking opportunities. The PhD candidate will enrol in MUV’s structured PhD program “Vascular Biology”, offering further networking opportunities (e.g. Young Scientist Association) and access to various career development programs (e.g. PhD mentoring, CareerMosaic).
The project will be conducted in cooperation with national and international collaboration partners at Umeå University (SE), University of Birmingham (UK), University of Sargodha (PAK), and Medical University of Innsbruck (AT), providing opportunities for laboratory rotations abroad if beneficial for the project and desired by the PhD student.
• Master’s degree (or equivalent) in biomedical sciences or a related discipline (e.g. molecular biology, microbiology, genetics, cell biology, …) by autumn 2026 at the latest
• Strong interest in biomedical research, haemostasis and infection biology
• Strong hands-on experience in wet lab and standard molecular biology techniques
• Willingness to work with murine disease models, human blood and BSL2 pathogens is required
- Medical physics and/or Biophysics
- Molecular and cell biology
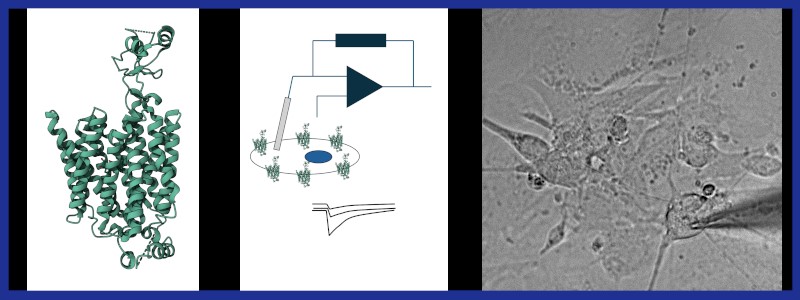
Organic cation transporters (OCTs) are found in almost every tissue and transport a variety of positively charged substrates ranging from neurotransmitters to toxins and drugs. The three isoforms (OCT1-3) differ in terms of pharmacological characteristics and expression pattern, with OCT1 mostly found in the liver, OCT2 in the kidney, and OCT3 in the heart and nervous system, where the latter contributes to low-affinity/high-capacity reuptake of monoamines and has been proposed as novel target for antidepressant therapy.
Electrophysiological experiments revealed a pronounced voltage dependence of Vmax and substrate KM values for rat OCT1&2. Such data are missing for OCT3, even though the latter is expressed in excitable cells, and therefore its voltage dependence of function must be expected to be physiologically relevant.
In this project, first a comprehensive biophysical characterization of human (i) wild-type and (ii) naturally occurring genetic variants of OCT3 expressed in HEK293 cells will be obtained. Substrate-induced currents through these transporters will be measured by whole-cell patch-clamp and for direct comparison, human OCT1&2 will be subjected to akin experiments.
In a second step, the physiological impact of OCT3 voltage dependence will be tested in co-cultures of rat sympathetic neurons and cardiomyocytes. The change in cardiomyocyte contraction frequency via sympathetic neuron stimulation will be measured, by triggering neuronal action potentials at increasing frequencies, in the absence and presence of OCT3 inhibitors. This allows to measure the relative contribution of OCT3 to elimination of norepinephrine at the synaptic level at different presynaptic potentials.
Skills to be acquired during PhD training
During the PhD project, the student will gain comprehensive training in cellular electrophysiology, membrane transport biology, and quantitative analysis of transporter function, including:
- Electrophysiological characterization of membrane transporters using whole-cell patch-clamp recordings
- Investigation of voltage-dependent transport mechanisms in organic cation transporters (OCTs)
- Generation and maintenance of mammalian cell culture models, including HEK293 expression systems and primary cell cultures
- Establishment of co-culture systems of sympathetic neurons and cardiomyocytes to study neurotransmitter transport and synaptic regulation
- Application of live-cell microscopy to monitor cellular and physiological responses
- Quantitative analysis and kinetic modelling of transporter activity
- Integration of electrophysiological, cellular, and physiological data to understand transporter function in excitable tissues
- Development of scientific communication, experimental design, and data interpretation skills
PhD network and research environment
The project is embedded in the Molecular Signal Transduction PhD Program at the Medical University of Vienna.
The program provides:
- A structured doctoral training environment focused on molecular and cellular signaling mechanisms
- Access to state-of-the-art research infrastructure and methodological expertise across participating laboratories
- Regular seminars, training modules, and workshops covering advanced research methods, scientific communication, and career development
- Opportunities for interdisciplinary exchange and collaboration within a vibrant doctoral student community
- Participation in international scientific meetings and networking with leading researchers in the field
Master’s degree (or equivalent) in biology, biochemistry, molecular biology, physiology, neuroscience, biomedical sciences, or a related field
Advantage: Experience in patch-clamp
- Human research
- Mental health
- Molecular and cell biology
- Public health
Hemophilia A and B are rare inherited bleeding disorders caused by X-linked mutations in the F8 and F9 genes. Over recent years, the therapeutic landscape of hemophilia has evolved substantially, ranging from non-factor therapies and extended half-life factor concentrates to gene therapy, which has already been recently administered at our center for the first time in Austria.
Despite these impressive advances and a significant reduction in bleeding complications, complete eradication of joint bleeds and subsequent development of debilitating hemophilic arthropathy has not yet been achieved and remains a major clinical challenge. Important unanswered questions persist regarding the role of subclinical joint bleeds, the underlying pathophysiology of hemophilic joint disease, and the observed heterogeneity in bleeding phenotypes among people with comparable residual endogenous factor levels.
While life expectancy for people with hemophilia has improved considerably, it still lags behind that of the general population, and health-related quality of life remains substantially impaired. In addition, a direct comparison of the hemostatic effect mediated by the different types of recently approved non-factor therapies with established factor concentrates, whose activity can be readily measured using routine coagulation assay, continuous to be an elusive goal.
This PhD project will build on the expanding range of therapeutic options in hemophilia, as well as data from an established nationwide registry on inherited bleeding disorders, the Austrian Hemophilia registry (https://bluter.at/wp/osterr-hamophilie-register) and associated biobanks. The specific research focus will be developed collaboratively with the selected candidate, taking into account their scientific interests.
Skills to be acquired during the PhD
- In-depth understanding of inherited bleeding disorders, with a particular focus on hemophilia A and B
- Experience working with a large, prospective nationwide registry, including patient characterization, longitudinal follow-up, and integration of clinical data
- Expertise in assessing joint health in people with hemophilia using both clinical joint scores (HJHS) and ultrasound evaluation (HEAD-US)
- Proficiency in conducting pharmacokinetic studies using population pharmacokinetics tools (e.g., WAPPS-Hemo, https://www.wapps-hemo.org)
- Expertise in the handling and processing of human samples, including plasma, platelets, and blood cells
- Advanced laboratory skills, including thrombin generation, plasmin generation, and clot formation and lysis assays
- Experience in biomarker discovery and correlation with clinical outcomes
- Competence in data analysis, statistical modelling, and interpretation of complex clinical datasets
- Well-developed scientific communication skills, including manuscript preparation, presentations, and interdisciplinary collaboration
- Experience working in a translational research environment bridging clinical medicine and molecular biology
PhD network and research environment
The candidate will joint an interdisciplinary team of clinicians and biologists dedicated to improving the care of people with inherited bleeding disorders, with a particularly focus on hemophilia. They will be closely integrated into the hemophilia team and actively contribute to clinical and research activities, including conducting pharmacokinetic studies and assessing joint health using clinical scoring systems and ultrasound.
A central component of the PhD project will involve the maintenance and analysis of the Austrian Hemophilia registry, in which patient data have been prospectively collected since 2009. The registry is complemented by a comprehensive biobank, quality of life questionnaires, data from pharmacokinetic studies, and patient infusion diaries, both paper-based as we all as electronic.
In addition, we offer numerous opportunities to participate in translational research within our two laboratories. Our research group is embedded in a strong international network, providing access to collaborations and enabling the candidate to gain valuable international research experience throughout the course of the project.
Medical Degree (equivalent to the Austrian Medical Doctors degree)
Advantageous: hands-on experience with general laboratory methods, including coagulation assays
- Animal research
- Cancer
- Cardiovascular systems
- Drug targets and/or drug development
- Human research
- Molecular and cell biology
- NET‑IT DocFund
Acquired mutations in aging hematopoietic stem cells (HSC) influence fitness and result in transition from polyclonal to monoclonal blood production. This condition is called clonal hematopoiesis (CH). Frequency of CH in the population increases with age (10-30%, >70 years). The most common genes somatically mutated in CH include DNMT3A, TET2, SRSF2, SH3B1 and the tyrosine kinase JAK2. These mutations not only allow the clonal dominance of the mutated HSCs but also impact the function of terminally differentiated myeloid cells produced by mutant HSCs. Recent studies have shown that individuals with CH have increased risk for atherosclerotic cardiovascular disease (CVD). Blood cells in CH may preferentially infiltrate atherosclerotic lesions, produce more chemokines than non-mutated cells and display inflammatory phenotype. We hypothesize that the altered function of the mutated myeloid cells in CH may be a significant source of inflammatory and prothrombotic stimuli that lead to disease development. We will characterize the functional impact of CH-associated mutations on monocyte, neutrophil, platelet function with a particular focus on innate immune signaling, cytokine production, activation/suppression, neutrophil-extracellular trap formation and coagulation. We will study the inflammatory features of mutated myeloid cells in transgenic murine models. Using bone marrow transplantation, we will introduce variable numbers of genetically tagged mutant myeloid cells into Ldlr-/- recipients and assess inflammatory phenotype and the impact on aortal lesion formation in a murine atherosclerosis model. Understanding the pathogenetic mechanisms triggered by CH in different disease contexts will have significant impact on patient management and open possibilities for therapeutic intervention.
Skills to be acquired during PhD training
During the PhD project, the student will gain interdisciplinary training in hematopoiesis, immunology, and cardiovascular disease research, including:
- Working with transgenic mouse models and experimental models of atherosclerosis
- Application of bone marrow transplantation techniques to study clonal hematopoiesis in vivo
- Functional characterization of monocytes, neutrophils, and platelets
- Investigation of innate immune signaling pathways and cytokine production
- Analysis of neutrophil extracellular trap (NET) formation and inflammatory responses
- Investigation of coagulation pathways and thrombo-inflammatory mechanisms
- Molecular and cellular analysis of disease phenotypes in experimental models
- Integration and interpretation of immunological and cardiovascular disease data
- Development of scientific communication and interdisciplinary collaboration skills
PhD network and research environment
The project is part of the FWF DocFund NET-IT (Dissecting the network of vascular inflammation and thrombosis). As a NET-IT PhD student, the candidate will join a doctoral college bringing together 10 research groups and a cohort of PhD students working on complementary aspects of vascular inflammation, thrombosis, and infection. The program provides an interdisciplinary setting that encourages scientific exchange and collaboration across laboratories.
The network provides:
- Internal and international laboratory rotations within partner research groups
- Structured training modules with yearly thematic focuses
- Courses on research methods, soft skills, grant writing, and scientific publishing
- Opportunities for interaction and collaboration with researchers from academia and industry
Master degree in biology (M.Sc.) or Medical degree (M.D.)
- AI/ML
- Bioinformatics
- Biomedical engineering
- Cardiovascular systems
- Computer lab
- Mathematics and/or Statistics
- Public health
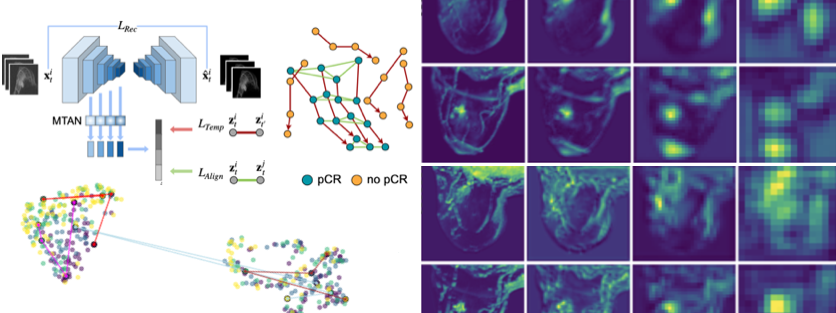
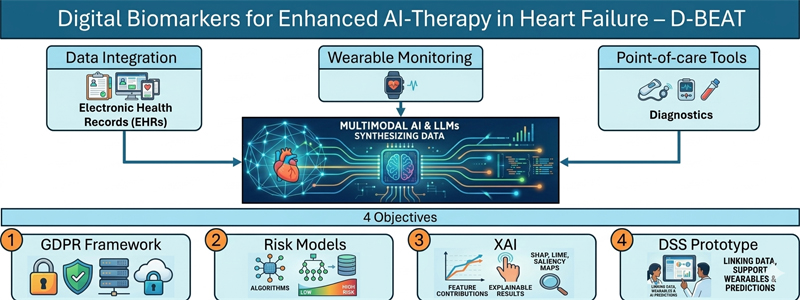
Heart failure (HF) care is moving toward earlier detection and personalized therapy. Artificial intelligence (AI), including large language models (LLMs), can synthesize data from electronic health records, wearables, and point-of-care tools to enable continuous clinical decision support across the full spectrum of HF severity. Our group has shown that digital biomarkers derived from patients with a blood pump implanted can predict complications including thrombosis, arrhythmias, infection, and volume imbalance.
We hypothesize that combining structured clinical data, wearable monitoring, and multimodal AI will enable early prediction of adverse events and optimized therapy in cardiovascular patients. The project pursues four objectives: (1) establishing a GDPR/MDR-compliant data integration framework for HF patients (NYHA II–IV); (2) developing LLM- and Machine Learning-based risk stratification models validated on institutional datasets; (3) implementing explainability methods (e.g. SHAP, LIME, saliency maps) for transparent clinical outputs; and (4) building a prototype decision-support tool linking institutional data, wearables, and AI algorithms.
The platform will integrate hospital, wearable, and point-of-care data to deliver early warnings, personalized diagnostics, and therapy recommendations. Explainability features will support clinician trust and enable integration into cardiology workflows, reducing complications and hospitalizations.
Skills to be Acquired During PhD Training
During the PhD project, the candidate will receive interdisciplinary training at the intersection of clinical cardiology, data science, and biomedical engineering. The training will include:
- Design and implementation of multimodal AI for clinical time-series and electronic health record data
- Integration and harmonization of heterogeneous data sources including electronic health records, wearable sensors, and point-of-care devices
- Application of explainable AI methods to ensure clinical interpretability and actionable model outputs
- Development of GDPR- and MDR-compliant data pipelines for sensitive medical data in real-world healthcare settings
- Prototype software development linking AI algorithms to clinical decision-support workflows in cardiology and cardiac surgery
- Statistical analysis, validation methodology, and critical evaluation of predictive model performance
- Scientific writing, presentation of research findings, and communication in an international research environment
- Translational research skills linking algorithmic outputs to clinical practice and patient outcomes
PhD Network and Research Environment
This PhD position is part of the Ludwig Boltzmann Institute for Cardiovascular Research, offering a collaborative and interdisciplinary research environment. It will be conducted at the Center for Medical Physics and Biomedical Engineering at the Medical University of Vienna with internationally recognized expertise in cardiovascular medicine and digital health. The candidate will be embedded in a multidisciplinary team combining cardiologists, data scientists, and biomedical engineers, with direct access to patient data and institutional datasets from a high-volume tertiary care center.
The position is integrated into a structured doctoral program offering dedicated training in research methods, biostatistics, scientific communication, and AI in medicine. The candidate will benefit from participation in national and international collaborations in cardiovascular AI research, access to cutting-edge computational infrastructure and clinical data platforms, and close mentorship from both clinical and engineering supervisors. Active involvement in scientific conferences and publication activities is expected and supported throughout the PhD.
The candidate will benefit from regular meetings, seminars, journal clubs, and retreats. These interactions provide excellent opportunities to learn new methods, exchange ideas, and build a strong professional network, supporting both your scientific and career development.
• Master's degree in biomedical engineering, computer science, data science, electrical engineering, or a related discipline with a quantitative focus
• Strong programming skills, particularly in Python, with experience in machine learning frameworks
• Experience with data processing, statistical analysis, and working with real-world datasets
• Strong analytical thinking, scientific curiosity, and the ability to work independently in a structured research environment. Very good English communication skills.
Advantage:
• Experience with clinical or medical data (electronic health records, ECG, wearable data)
• Familiarity with explainable AI methods and model interpretability tools
• Knowledge of regulatory frameworks for medical software (GDPR, MDR) or interest in acquiring this knowledge
• Prior experience in a clinical or translational research setting
- Animal research
- Bioinformatics
- Cardiovascular systems
- Human research
- Immunology
- Molecular and cell biology
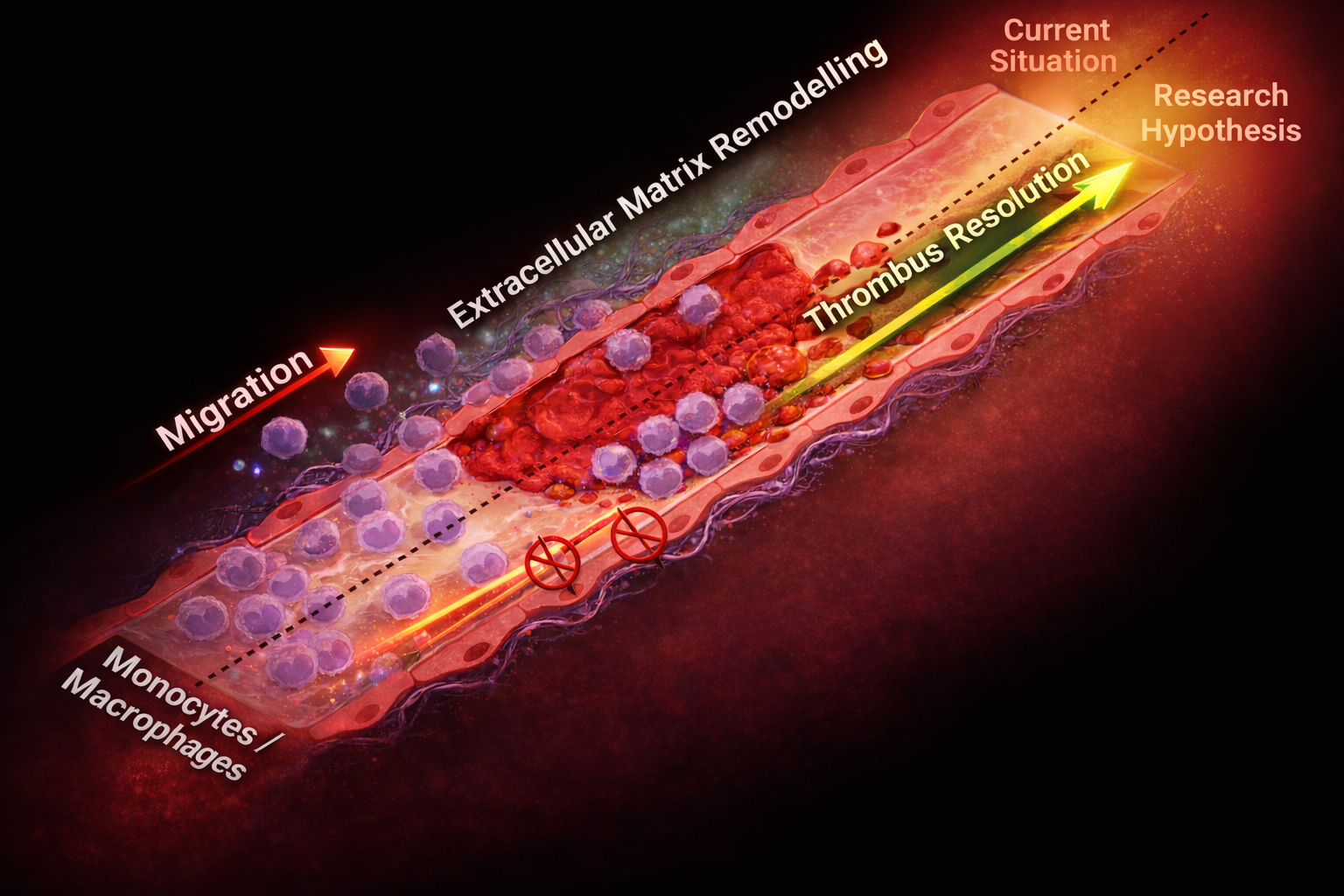
Venous thrombosis is a major and increasing health burden, with deep-vein thrombosis often leading to long-term complications such as post-thrombotic syndrome, chronic pain, venous hypertension, and ulcers. Although existing treatments focus on reopening the vessel, they do not specifically address the damage inflicted on the venous endothelium. We are looking for a highly motivated PhD candidate to join us in studying how endothelial protection and mechanical stress shape thrombus resolution and long-term vein-wall remodeling.
Our work has shown that reduced monocyte infiltration accelerates thrombus resolution by preventing early thrombus stabilization. We further found that monocytes migrate along the endothelium and cross the endothelial barrier to remodel the vessel wall. Building on these findings, this project will investigate whether strengthening endothelial junctions can redirect monocyte and macrophage migration, reduce fibrosis, and improve long-term venous function. At the same time, we will address how thrombus-induced mechanical stress reprograms the venous endothelium and promotes a persistent pro-thrombo-inflammatory state.
To answer these questions, you will use mouse models of venous thrombosis, in vivo thrombotic challenge experiments, advanced microscopy, flow cytometry, single-cell epigenomic approaches, and proteomics, combined with translational analyses of human material working closely with the Department of Angiology, headed by Prof. Sabine Steiner. You will work in an interdisciplinary environment linking vascular biology, thrombosis research, and clinical translation.
By the end of the project, we aim to understand how preserving endothelial barrier integrity alters clot resolution and vein-wall remodeling, and how mechanical stress leaves a lasting imprint on endothelial function. These findings will provide the basis for new translational strategies to prevent long-term complications after venous thrombosis.
Skills to be acquired during PhD training
- Application of mouse models of venous thrombosis, including vena cava ligation and in vivo thrombotic challenge experiments.
- In vivo testing of endothelial barriermodulating strategies.
- Analysis of monocyte and macrophage migration patterns during thrombus resolution and vein-wall remodeling.
- Assessment of thrombus resolution, vessel wall integrity, fibrosis, and recurrence risk using histological and imaging-based approaches.
- Generation and interpretation of single-cell and epigenomic datasets, including ATAC-seq, methylation profiling, and proteomics.
- Bioinformatics skills for histology assessment and quantitative omics data analysis, including integration of murine and human datasets
PhD network and research environment
This PhD project is embedded in the Ludwig Boltzmann Institute for Cardiovascular Research, offering a strongly interdisciplinary and translational research environment at the interface of vascular biology, thrombosis research, and clinical medicine. The project benefits from close collaboration with the Helmholtz Institute for Metabolic, Obesity and Vascular Research (HI-MAG) in Leipzig, providing access to complementary expertise and an extended international research network in vascular and translational science. The PhD candidate will further benefit from the LBG Career Center, which offers career-development support through workshops, events, and dedicated training formats. In addition, the project includes the possibility for international laboratory rotations as well as participation in external and specialized training modules, ensuring broad scientific and professional development throughout the PhD.
- Animal research
- Cardiovascular systems
- Immunology
- Molecular and cell biology
- NET‑IT DocFund
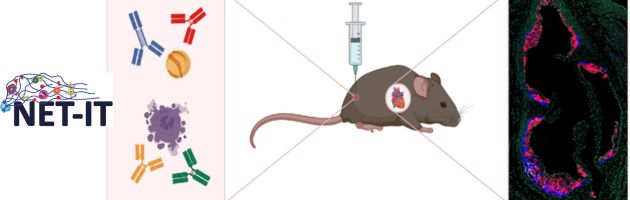
Atherosclerotic cardiovascular disease is the leading cause of death globally. It is driven by the accumulation of cholesterol-carrying lipoproteins in the artery wall, which triggers an inflammatory response. A better understanding of the immune-mediated processes involved may help identify novel therapies to treat this disease. B cells and humoral immunity play an important role in this process, particularly through antibodies targeting oxidation-specific epitopes (OSE), which are generated during lipid peroxidation of these lipoproteins. However, the role of OSE-specific IgG is poorly understood. The aim of this project is to dissect the effector functions of OSE-specific IgG antibodies in atherogenesis. This will be done by combinig functional assays evaluating the impact of IgG on immune and stromal cells in vitro and in vivo studies to investigate the impact in various preclinical models of atherosclerosis. Antibody-mediated effects on atherosclerosis will be elucidated by integrating data from multiple omics-based analyses with immunological, histological and molecular profiling of pathophysiological changes in these models. This project will ultimately clarify IgG-specific mechanisms in atherosclerosis and may pave the way for novel OSE-targeted therapeutic strategies in cardiovascular disease.
Skills to be Acquired During PhD Training
During this PhD project, the student will gain interdisciplinary training in immunology, cardiovascular biology, and systems-level data analysis, including:
- Design and execution of functional immunological assays to investigate antibody-mediated cellular responses
- In vitro studies of immune and vascular cell interactions, including macrophage, endothelial and smooth muscle cell biology
- Application of preclinical models of atherosclerosis to study disease mechanisms in vivo
- Analysis of antibody effector functions and their impact on inflammation and tissue remodeling
- Integration of multi-omics datasets (e.g., transcriptomics, proteomics) with immunological and histological data
- Advanced molecular and histological techniques for tissue characterization
- Development of scientific communication, interdisciplinary collaboration, and critical data interpretation skills
PhD network and research environment
The project is part of the FWF DocFund NET-IT (Dissecting the network of vascular inflammation and thrombosis). As a NET-IT PhD student, the candidate will join a doctoral college bringing together 10 research groups and a cohort of PhD students working on complementary aspects of vascular inflammation, thrombosis, and infection. The program provides an interdisciplinary setting that encourages scientific exchange and collaboration across laboratories.
The network provides:
- Internal and international laboratory rotations within partner research groups
- Structured training modules with yearly thematic focuses
- Courses on research methods, soft skills, grant writing, and scientific publishing
- Opportunities for interaction and collaboration with researchers from academia and industry
- Access to state-of-the-art infrastructure for immunology, cardiovascular research, and omics-based analyses
- Opportunities to collaborate with research groups working on biomarkers, inflammation, and cardiovascular disease mechanisms
Master/Diploma’s degree in Medicine, Biology, Molecular Biology, Biotechnology, or a related subject.
Advantage: Experience in mouse-based research
- Cancer
- Computer lab
- Human research
- Immunology
- Molecular and cell biology
- Wet lab
How environmental exposures reshape the architectural and physical microenvironment of human skin and influence melanoma progression remains poorly understood. This PhD project aims to investigate how ultraviolet radiation and environmental chemicals alter the extracellular matrix (ECM) and thereby affect melanoma cell behaviour. The project will develop advanced experimental models to study ECM remodelling in human skin. The candidate will establish human skin exposure models and organotypic 3D dermis models to investigate how environmental factors influence ECM structure, mechanics, and melanoma cell-matrix interactions. These systems preserve native tissue architecture and enable controlled experimental manipulation of environmental conditions. A central component of the project will be the biophysical and structural characterisation of dermal ECM. Using advanced microscopy techniques, including multiphoton and second harmonic generation microscopy, the candidate will analyse collagen fibre organisation, matrix alignment, ECM remodelling and cell-matrix interactions. These analyses will be complemented by mechanical measurements such as rheology and other biophysical approaches to determine tissue stiffness, viscoelastic properties and the forces exerted by cells on the surrounding matrix. The project is embedded in a multidisciplinary and international consortium combining cell biology, advanced imaging, biophysics, computational modelling and clinical research to better understand how environmental exposures reshape the melanoma microenvironment and contribute to tumor onset and progression.
Skills to be acquired during PhD training
During the PhD project, the student will gain interdisciplinary training in cancer biology, advanced microscopy, and biophysical analysis, including:
- Development of organotypic 3D skin and dermis models
- Establishment of ex vivo human skin exposure assays
- Advanced 2D and 3D microscopy techniques, including:
- Confocal microscopy
- Multiphoton microscopy
- Second harmonic generation (SHG) imaging
- Brillouin microscopy
- FRAP (fluorescence recovery after photobleaching)
- Quantitative microscopy and image analysis using MATLAB and ImageJ/Fiji
- Biophysical measurements of cell and tissue stiffness
- 3D cell migration assays and analysis of cell–matrix interactions
- Molecular biology and transcriptomic approaches including qPCR and next-generation sequencing
- Integration of imaging, mechanical, and molecular datasets to understand tumor microenvironment dynamics
PhD Network and Research Environment
The PhD project will be conducted in a multidisciplinary and international research consortium focusing on melanoma biology and the tumor microenvironment.
The PhD student will benefit from:
- A collaborative environment combining cell biology, biophysics, imaging, computational modelling, and clinical research
- Access to state-of-the-art microscopy platforms and biophysical instrumentation
- Training opportunities in advanced imaging technologies and quantitative analysis
- Opportunities to participate in translational cancer research and interdisciplinary collaborations
- Close interaction with researchers across basic science and clinical disciplines
Candidates should ideally have a background in molecular biology, cell biology, or a related field. Applicants with a medical background (medical graduates or MD/PhD students) are encouraged to apply if they have a strong interest in experimental (wet-lab) research and dermatology
• Experience with cell culture and/or in vitro model systems
• Experience with microscopy techniques, preferably confocal microscopy
• Basic molecular biology skills (e.g. PCR, Western blotting)
• Strong interest in cell-matrix interactions, cancer biology, and imaging-based research
• Ability to work independently and collaboratively in an interdisciplinary research environment
Advantage
• Experience with organotypic or 3D cell culture models
• Experience with advanced 2D or 3D imaging techniques and quantitative image analysis
• Background in biophysics, bioengineering, or quantitative imaging
• Interest in translational research and patient engagement (optional opportunities to participate in clinical activities)
- Biomedical engineering
- Cardiovascular systems
- Wet lab
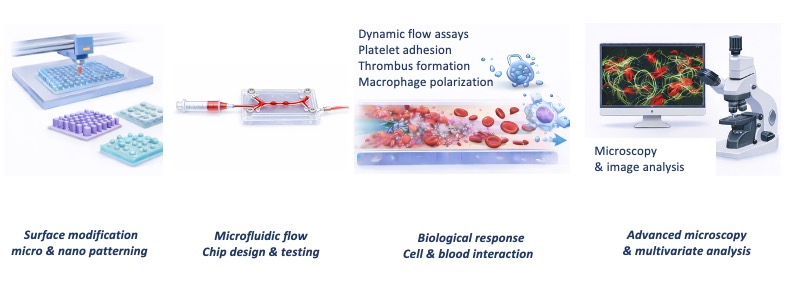
The hemocompatibility of cardiovascular implants is critically determined by their surface properties, which modulate protein adsorption, platelet adhesion, immune activation, and thrombus formation. Recent advances in nano- and microscale engineering have enabled bioinspired surface topographies with anti-fouling and superhydrophobic characteristics that significantly reduce platelet interaction under static conditions. However, their functional stability and biological performance under physiologically relevant flow conditions remain poorly understood. This PhD project will develop robust experimental platforms to systematically investigate how microstructured and functionalized surfaces influence blood and immune responses under dynamic conditions. The candidate will design, fabricate and optimize microstructured surfaces using advanced additive manufacturing techniques and may apply functional coatings (e.g., pSBMA or nitric oxide donors) to further enhance hemocompatibility. Dynamic flow assays will be developed to study platelet adhesion, activation, and thrombus formation under controlled shear conditions. In parallel, the project will investigate the immunomodulatory effects of surface topography, focusing on M1 and M2 macrophage polarization and its interplay with platelet behavior. Quantitative data on surface structure, wettability, and biological responses will be integrated using multivariate statistics and machine learning to establish predictive models that correlate design parameters with hemocompatibility outcomes. This interdisciplinary project offers comprehensive training in biomaterials surface engineering, microfluidic design, cell–biomaterial interactions, dynamic flow assay development, advanced microscopy, and data-driven analysis. The results will contribute to the rational design of next-generation blood-contacting medical devices and provide mechanistic insights into surface–cell interactions under physiologically relevant conditions.
Skills to be acquired during PhD training
During the PhD project, the student will gain interdisciplinary training in biomaterials engineering, cardiovascular device research, and quantitative analysis of biomaterial–cell interactions, including:
- Design and fabrication of micro- and nanostructured biomaterial surfaces using advanced additive manufacturing techniques
- Functionalization of implant surfaces with bioactive coatings to enhance hemocompatibility
- Development of microfluidic systems and dynamic flow chambers for physiologically relevant in vitro assays
- Design and execution of static and dynamic blood–material interaction assays, including platelet adhesion and thrombus formation studies
- Investigation of immune responses to biomaterials, including macrophage polarization and cell–biomaterial interactions
- Application of advanced microscopy techniques and quantitative image analysis
- Integration of biological and material parameters using multivariate statistics and machine learning approaches
- Development of scientific communication, interdisciplinary collaboration, and data interpretation skills
PhD network and research environment
The PhD position is embedded in the collaborative research environment of the Ludwig Boltzmann Institute for Cardiovascular Research.
The PhD student will benefit from:
- Access to state-of-the-art infrastructure for biomaterials research, microfabrication, and cardiovascular biology
- A collaborative and interdisciplinary research environment involving experts in biomaterials engineering, cardiovascular medicine, and device development
- Regular scientific meetings, seminars, journal clubs, and retreats within the institute
- Opportunities to collaborate with additional research groups working on biomarkers, cardiovascular remodeling, and translational cardiovascular research
- A supportive network promoting scientific exchange, interdisciplinary learning, and professional development
• Master’s degree in biomedical engineering, materials science, bioengineering, or related field.
• Knowledge of biomaterials and cell–biomaterial interactions.
• Experience with cell culture and basic in vitro assays.
Advantage:
• Experience in micro/nanofabrication (3D printing) or surface functionalization.
• Familiarity with microfluidics or flow assays.
• Skills in advanced microscopy, immunological assays, or data analysis.

- Basic research
- Cardiovascular systems
- Human research
- Immunology
- Molecular and cell biology
- NET‑IT DocFund
- Wet lab
Thrombosis and inflammation are tightly interconnected processes that contribute to many acute and chronic diseases. During infections or systemic inflammatory responses, the mechanisms driving thrombus formation differ fundamentally from classical injury-induced coagulation. In these settings, immune responses and host-derived signals profoundly reshape platelet function and can trigger a pathological form of clotting known as thromboinflammation. Increasing evidence suggests that platelets act as key regulators at the interface of immunity and coagulation by sensing inflammatory cues and interacting with immune cells to coordinate vascular responses. However, the mechanisms by which host responses during infection alter platelet behavior and promote thrombotic complications remain poorly understood.
Research Aim and Approach
This project aims to uncover how inflammatory host responses modulate platelet function and drive thromboinflammatory processes. We will investigate how immune-derived danger signals, fever, and changes in the physiological environment influence platelet activation and platelet–immune cell interactions. Using a multidisciplinary approach combining cellular immunology, vascular biology, advanced imaging, and in vivo disease models, we will identify key pathways linking immune activation to pathological clot formation.
By dissecting the mechanisms connecting infection, inflammation, and thrombosis, this research aims to distinguish protective haemostatic responses from harmful pathological clotting and to identify novel strategies to prevent thrombotic complications during severe inflammatory diseases.
Skills to be acquired during PhD training
During the PhD project, the student will acquire training in thromboinflammation research, platelet biology, and immune–vascular interactions, including:
- Understanding mechanisms of thromboinflammation and interactions between immune and vascular systems
- Application of experimental techniques such as flow cytometry, confocal microscopy, flow-based thrombosis assays, and functional immune cell assays
- Work with human samples and murine models of infection and inflammation
- Use of in vivo models of thrombosis
- Training in experimental design and data analysis
- Experience in interpreting complex biological datasets
- Development of scientific communication skills, including manuscript preparation, conference presentations, and grant writing
- Experience working in a collaborative and international research environment
- Development of transferable skills such as project management, teamwork, and interdisciplinary collaboration
PhD network and research environment
The project is part of the FWF DocFund NET-IT (Dissecting the network of vascular inflammation and thrombosis). As a NET-IT PhD student, the candidate will join a doctoral college bringing together 10 research groups and a cohort of PhD students working on complementary aspects of vascular inflammation, thrombosis, and infection. The program provides an interdisciplinary setting that encourages scientific exchange and collaboration across laboratories.
The network provides:
- Internal and international laboratory rotations within partner research groups
- Structured training modules with yearly thematic focuses
- Courses on research methods, soft skills, grant writing, and scientific publishing
- Opportunities for interaction and collaboration with researchers from academia and industry
Master in Natural or Medical Sciences or equivalent
Advantage: Experience with mouse models

- Epidemiology
- Mathematics and/or Statistics
- Public health
Women carrying germline BRCA1 or BRCA2 pathogenic variants (gBRCA1/2 PV) face substantially increased lifetime risks of breast cancer and often undergo risk-reducing salpingo-oophorectomy (RRSO) at a young age. Hormone replacement therapy (HRT) is frequently prescribed to manage premature menopausal symptoms, yet its impact on breast cancer risk in this high-risk population remains insufficiently understood and clinically controversial. This MSCA-Doctoral Network funded PhD project (DC4) aims to evaluate the association between HRT use and breast cancer risk and outcomes in BRCA1/2 pathogenic variant carriers, and to identify modifying factors that influence individual risk trajectories. The project will generate evidence to support personalized risk counselling and clinical decision-making in hereditary breast cancer care. The doctoral candidate will quantify the association between different HRT regimens and breast cancer risk and outcomes in gBRCA1/2 carriers, assess effect modification by non-genetic factors (e.g. BMI, lifestyle, clinical characteristics, etc.), integrate genetic and non-genetic risk factors into existing risk prediction frameworks (e.g. CanRisk), and contribute to improved, evidence-based recommendations for HRT use and risk-reducing strategies in hereditary breast cancer care.
Skills to be acquired during PhD training
- Designing and conducting epidemiological studies in high-risk populations, including cohort and registry-based analyses
- Statistical analysis of complex datasets using software such as R, Stata, or SAS, including survival analysis, regression modeling, and interaction assessment
- Integrating genetic and non-genetic risk factors into risk prediction frameworks (e.g., CanRisk)
- Critical appraisal of scientific literature and development of evidence-based clinical recommendations
- Communicating research findings effectively in scientific publications, conferences, and clinical settings
- Collaborating across disciplines, including genetics, epidemiology, and clinical oncology, to translate research into personalized patient care
PhD network & research enviroment
The PhD student will be part of the HER-CARE MSCA Doctoral Network, joining an international cohort of 15 PhD researchers working on hereditary and early-onset breast cancer across multiple institutions. The student will benefit from network-wide training, workshops, and mentoring in epidemiology, genetics, and clinical research. Additionally, as part of the MSCA-DN program, the student will undertake two planned international secondments: one at the University of Cologne (Dr. Karin Kast) and one at SOPHiA Genetics in Years 2 and 3, gaining complementary academic and industry experience and fostering cross-sectoral collaborations.
Applicants must meet the MSCA Doctoral Network eligibility criteria:
- A Master’s degree in epidemiology, biostatistics, genetics, biomedical sciences or equivalent by the start date of the PhD contract
- Experience in statistical analysis (e.g. R, Stata or SAS)
- Strong interest in hereditary cancer, women’s health and clinical research
- No PhD degree at the time of recruitment
- Mobility rule: candidates must not have lived, worked, or studied in the country of the recruiting beneficiary for more than 12 months in the 36 months prior to recruitment. Any nationality is eligible, provided the mobility rule is fulfilled
- Animal research
- Cardiovascular systems
- Human research
- Immunology
- NET‑IT DocFund
- Wet lab
Lung infections are a leading cause of death in the ageing population. At the same time, these people often suffer from underlying disorders like cardiovascular diseases (CVD) or clonal hematopoiesis. These chronic diseases are associated with a systemic pro-inflammatory and pro-coagulant state, two aspects that have been found to also affect local, tissue specific immune responses (such as in the lungs). However, the underlying mechanisms how these changes can impair the host defense against respiratory pathogens is poorly understood. Within this PhD project, we will examine lung-tissue specific host defense mechanisms and vulnerabilities in the context of chronic inflammatory and pro-coagulant conditions. To do so, we will study aged mice and selected “perturbations” associated with altered inflammation and coagulation profiles and examine the impact on pulmonary immune cell functionalities, and the host response to relevant bacterial and viral pathogens. The close interaction with the NET-IT community will enable access to all relevant models of cardiovascular diseases and clonal hematopoiesis, which together with our expertise in pulmonary host defense mechanisms will enable us to get deep insights into the molecular mechanisms of the known link between chronic disorders and the poor outcome from lung infections. This knowledge will form the basis for novel preventive or therapeutic approaches.
Skills to be acquired during PhD training
During this PhD project, the student will study how ageing and chronic diseases influence the body’s ability to respond to lung infections. The project combines infection biology and immunology and includes training in the following areas:
- Use of mouse models to study ageing, inflammation, coagulation, and respiratory infections
- Investigation of host defense mechanisms against bacterial and viral pathogens in the lung
- Analysis of pulmonary immune cell populations and their functional responses
- Application of immunological techniques such as flow cytometry and macrophage functional assays
- Use of single-cell transcriptomics (scRNA-seq) to analyze immune cell responses
- Study of how conditions such as cardiovascular disease and clonal hematopoiesis affect lung immunity
- Identification of molecular mechanisms linking chronic disease with increased susceptibility to lung infections
PhD network and research environment
The project is part of the FWF DocFund NET-IT (Dissecting the network of vascular inflammation and thrombosis). As a NET-IT PhD student, the candidate will join a doctoral college bringing together 10 research groups and a cohort of PhD students working on complementary aspects of vascular inflammation, thrombosis, and infection. The program provides an interdisciplinary setting that encourages scientific exchange and collaboration across laboratories.
The network provides:
- Internal and international laboratory rotations within partner research groups
- Structured training modules with yearly thematic focuses
- Courses on research methods, soft skills, grant writing, and scientific publishing
- Opportunities for interaction and collaboration with researchers from academia and industry
Master’s degree (or equivalent) in biomedical sciences or related fields.
Advantage: Experience with mouse models, flow cytometry or immunological assays.
- Bioinformatics
- Cardiovascular systems
- Computer lab
- Human research
- Immunology
- Molecular and cell biology
- NET‑IT DocFund
- Wet lab
Macrophages are versatile cells, fulfilling numerous functions including as a first line of defense during infection, tissue repair, and support of tissue integrity by recycling damaged mitochondria. To understand what functions is required, macrophages are sensitive to environmental cues and quickly adapt to fulfill the required task. These cues can be damage associated molecular patterns, pathogen associated proteins, cytokines or reactive oxygen species. But also the environment can trigger macrophage reactions including surface tension and mechanical forces. We are looking for someone interested to unravel the influence of those mechanical stresses on form and function of macrophages within atherosclerotic plaques with us. We expect that different areas of a plaque contain defined forces that alter macrophage function locally, thereby shaping macrophage reactivity towards additional signals. To help us unravel the underlying signals you will use in vitro techniques with primary human macrophages, 3D culture models and animal models of atherosclerosis. Experimental approaches will include spatial omics, confocal microscopy, flow cytometry, RNA sequencing, and metabolomics. At the end of your PhD thesis we want to understand how and through which pathways macrophages are shaped by their mechanical environment in an atherosclerotic plaque.
Skills to be acquired during PhD training
During the PhD project, the student will gain interdisciplinary training in immunology, vascular biology, and advanced molecular profiling techniques, including:
- Application of advanced wet-lab techniques to mechanically manipulate macrophages and study mechanobiological responses
- Use of primary human macrophages and 3D culture systems to model tissue environments in vitro
- Application of spatial omics technologies to identify and localize specific cell populations in human and mouse tissues
- Use of confocal microscopy and flow cytometry to analyze macrophage phenotype and function
- Generation and analysis of RNA sequencing and metabolomics datasets
- Experimental design and AI-supported analysis of high-dimensional sequencing data
- Use of in vivo mouse models of atherosclerosis
- Conducting translational research comparing findings from human and mouse tissues
- Development of scientific communication, data interpretation, and interdisciplinary collaboration skills
PhD network and research environment
The project is part of the FWF DocFund NET-IT (Dissecting the network of vascular inflammation and thrombosis). As a NET-IT PhD student, the candidate will join a doctoral college bringing together 10 research groups and a cohort of PhD students working on complementary aspects of vascular inflammation, thrombosis, and infection. The program provides an interdisciplinary setting that encourages scientific exchange and collaboration across laboratories.
The network provides:
- Internal and international laboratory rotations within partner research groups
- Structured training modules with yearly thematic focuses
- Courses on research methods, soft skills, grant writing, and scientific publishing
- Opportunities for interaction and collaboration with researchers from academia and industry
Master’s degree in molecular biology, cell biology, genetics, biomedical sciences or equivalent
Strong hands-on experience with wet laboratory techniques and an interest in data science and bioinformatics
Advantage: Basic knowledge of R
Note: This project will include the work with mouse models
- Cardiovascular systems
- Human research
- Immunology
- Molecular and cell biology
- NET‑IT DocFund
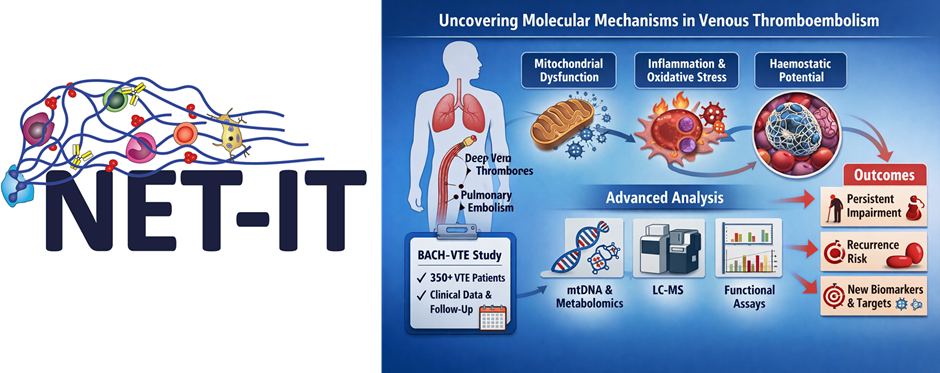
Venous thromboembolism (VTE), including deep vein thrombosis and pulmonary embolism, is a common and debilitating condition. Despite optimal treatment, a substantial proportion of patients experiences long-term functional impairment and reduced quality of life that cannot be explained by clinical factors alone. This suggests an underlying biological basis.
Emerging evidence highlights a close interplay between inflammation, metabolism, and coagulation. In particular, mitochondrial dysfunction and altered oxidative stress responses may contribute to persistent cellular changes following a thrombotic event. These alterations could promote a prothrombotic environment, impair thrombus resolution, lead to persisting functional limitations, and increase the risk of recurrence. However, the molecular mechanisms linking these processes to clinical outcomes remain poorly understood.
Aims and Approach
This project aims to identify molecular and cellular alterations in VTE patients that are associated with long-term functional impairment and thrombotic risk. Using samples and data from the prospective BACH-VTE cohort, the study will integrate clinical characterization with advanced molecular analyses.
The approach includes the assessment of mitochondrial function, mtDNA copy number, and oxidative stress, alongside global metabolomic profiling using LC-MS. In addition, haemostatic potential will be evaluated through functional assays such as thrombin generation, fibrin clot formation and lysis, extracellular vesicle activity, and neutrophil-extracellular trap (NET) formation. By linking these molecular signatures to clinical outcomes and VTE sequalae, the project seeks to improve understanding of VTE pathophysiology and identify novel biomarkers and therapeutic targets for better risk stratification and patient management.
Skills to be acquired during the PhD
- In-depth understanding of venous thromboembolism (VTE), thromboinflammation, and the interplay between inflammation, metabolism, and coagulation
- Experience working with a large prospective clinical cohort, including patient characterization, longitudinal follow-up, and clinical data integration
- Expertise in handling and processing human samples (plasma, platelets, blood cells)
- Advanced laboratory skills including thrombin generation assays, clot formation and lysis assays, extracellular vesicle analysis, and NET measurements
- Training in mitochondrial biology, including assessment of mtDNA copy number, mitochondrial function, morphology, and reactive oxygen stress
- Skills in biomarker discovery and correlation with clinical outcomes
- Competence in data analysis and interpretation of complex clinical and molecular datasets
- Development of scientific communication skills, including manuscript writing, presentations, and interdisciplinary collaboration
Experience working in a translational research environment bridging clinical medicine and molecular biology
PhD network and research environment
The project is part of the FWF DocFund NET-IT (Dissecting the network of vascular inflammation and thrombosis). As a NET-IT PhD student, the candidate will join a doctoral college bringing together 10 research groups and a cohort of PhD students working on complementary aspects of vascular inflammation, thrombosis, and infection. The program provides an interdisciplinary setting that encourages scientific exchange and collaboration across laboratories.
The network provides:
- Internal and international laboratory rotations within partner research groups
- Structured training modules with yearly thematic focuses
- Courses on research methods, soft skills, grant writing, and scientific publishing
- Opportunities for interaction and collaboration with researchers from academia and industry
Master in Natural or Medical Sciences, Medical Doctor
Advantage: Hands-on experience with general laboratory methods, including cell isolation techniques and coagulation assays

- Biochemistry
- Bioinformatics
- Cardiovascular systems
- Drug targets and/or drug development
- Endocrinology and Metabolism
- Immunology
- Molecular and cell biology
- Organ failure and/or replacement
- Wet lab
The recently opened Christian Doppler Laboratory for Peritoneal Regulation of the Immune-Metabolic Ecosystem in Peritoneal Dialysis (CDL-PRIME-PD) will conduct high-level, application-oriented basic research. The lab investigates metaflammation and immunometabolism to uncover novel therapeutic targets for patients with end-stage kidney disease, with a strong focus on peritoneal dialysis (PD). The project aims to elucidate the systemic impact of the metabolic burden during PD on other organs, particularly its role in driving metaflammation and cardiovascular complications. The successful candidate will apply cutting-edge experimental approaches, including in vivo disease models, metabolic imaging, metabolomics, proteomics, microbiome profiling, and integrative multi-omics analyses.
Skills to be acquired during PhD training
During the PhD project, the student will acquire interdisciplinary training in experimental biomedical research with a focus on immunometabolism and kidney disease. The training will include:
- Advanced wet-lab techniques for studying immune and metabolic processes in kidney disease and dialysis
- Use of in vivo disease models and experimental approaches to investigate systemic metabolic and inflammatory responses
- Application of high-throughput technologies such as metabolomics, proteomics, and microbiome profiling
- Analysis and integration of multi-omics datasets using biostatistics and bioinformatics tools
- Experimental design, data interpretation, and critical analytical thinking
- Scientific writing and presentation of research findings in an international research environment
- Experience in translational research aimed at identifying potential therapeutic targets for dialysis-related complications
PhD Network and Research Environment
The PhD project will be conducted at the Medical University of Vienna, an internationally recognized center for biomedical research. The student will be embedded in a multidisciplinary research environment combining expertise in immunology, metabolism, and kidney disease, with close interaction between basic scientists and clinicians.
PhD training includes participation in structured doctoral programs, seminars, and specialized courses in research methods, statistics, and scientific communication. The university provides access to state-of-the-art core facilities and promotes interdisciplinary collaboration and international scientific exchange.
Master’s degree in life sciences, natural sciences, or a related discipline (e.g., molecular biology, biochemistry, biotechnology, genetics, biomedical sciences).
Strong hands-on experience with wet laboratory techniques, preferably in molecular and/or cellular biology, and an interest in data science, particularly biostatistics and bioinformatics.
Experience working with mouse models is considered an advantage; however, willingness to work with mouse models is required.
- Cancer
- Drug targets and/or drug development
- Human research
- Immunology
- Molecular and cell biology
- Wet lab
Emerging evidence suggests that therapy resistant malignant cells arise from non-genetic mechanisms influenced by interactions with the tumor microenvironment (TME). We have developed highly advanced complex patient-derived tumor organoids (PDTOs) integrating autologous cancer-associated fibroblasts (CAFs) and immune cells that vividly mimic patient tissue characteristics. Within this project, we aim to use these highly complex patient-derived models to investigate how the TME is mediating response and resistance to gold-standard chemotherapy (ChT) treatment. We hypothesize that early molecular changes induced by ChT are shaped by the TME and enhance tumor-cell survival ultimately leading to acquired resistance. We aim to monitor early responses to ChT at the transcriptome and phospho-proteome level and compare responses within ChT-sensitive and resistant organoids. Additionally, we will conduct genome-wide CRISPR and functional perturbation screens (CROP-seq) to discover targetable susceptibility genes in ChT-R tumor cells, enabling identification of new drug targets that will be further tested in primary-material-derived tissue slice cultures and mice.
Skills to be acquired during PhD training
During the PhD project, the student will gain comprehensive training in translational biomedical research and advanced molecular technologies, including:
- Handling of primary human tissue and generation of advanced culture models
- Use of advanced sequencing technologies
- Analysis of next generation sequencing data
- Experience working in a collaborative, consortium-based research environment
- Development of scientific communication, data interpretation, and interdisciplinary collaboration skills
PhD network and research environment
The project is part of the CRC-Res Clinical Research Group, funded by the Ludwig Boltzmann Society.
The network includes seven research groups based in Vienna and promotes strong collaboration between basic scientists and clinicians.
PhD students benefit from:
- Close interaction with multiple research groups within the CRC-Res consortium
- An interdisciplinary research environment that promotes exchange between different scientific fields
- Opportunities for collaborative projects and knowledge sharing within the network
- A strong focus on translating basic research findings into clinically relevant applications
Master’s in Molecular Biology, Cellular Biology, Molecular Medicine or similar
Advantage: Experience with primary human material cell culture, potentially also working with human organoids
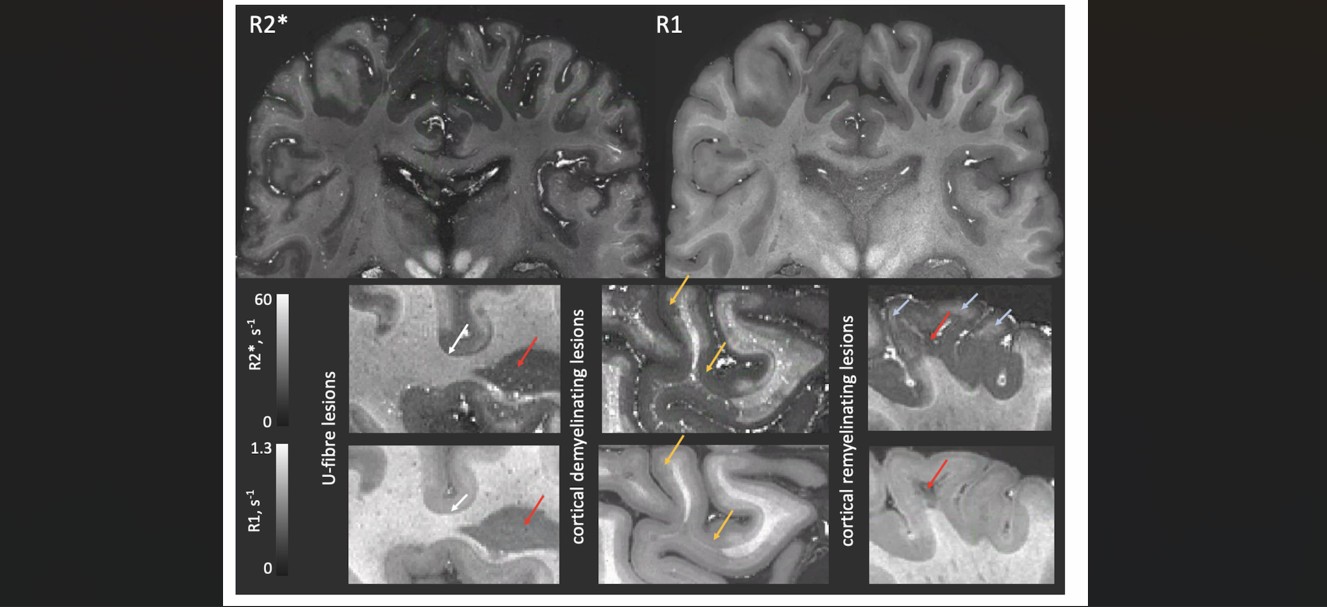
- Bioinformatics
- Biomedical engineering
- Human research
- Immunology
- Mathematics and/or Statistics
- Neuroscience
- Wet lab
Multiple sclerosis (MS) is the most common cause of acquired, non-traumatic disability in young people. Previously viewed primarily as an inflammatory disease of the brain's white matter, it is now considered a complex disorder involving focal and diffuse pathological processes throughout the brain, including the cerebral cortex. Infiltration of inflammatory cells, activation of phagocytic cells in the brain, and subsequent demyelination (loss of myelin sheaths that ensure nerve conduction velocity in the brain) occur throughout the central nervous system, including the gray matter, and shape the symptoms of affected individuals over time. Cortical pathology in MS is inherently difficult to detect with MRI, but very important for the patients due to the sometimes widespread affection of cortex in MS, the high variability between patients and the important cognitive consequences related to cortical pathology.With this project, we aim to utilize additional MRI tissue contrasts (MPM sequence), such as those derived from brain iron and myelin, by examining tissue samples from the brains of deceased individuals with MS in modern, high-resolution 7-Tesla MRI scanners and employing the latest sequence combinations. This will improve the visualization of myelin in the cerebral cortex. We believe this will also enhance the visualization of protective remyelination in the cerebral cortex in living subjects, enabling the testing of new therapies.
Skills to be acquired during PhD training
Magnetic resonance imaging, computational analysis of MRI data and histopathology with several software tools - QuPath,
PhD network and research environment
This project is part of a tri-national consortium with Nik Weiskopf / Evgeniya Kirilina from the Max Planck Institute in Leipzig, GER and Pierre Maquet / Emilie Lommers from CHU Liege Neurology, BEL. Options for visits, research exchange abroad!
Master’s degree in biomedical sciences or biology or medicine or (biophysical) engineering
Advantageous experience (optional):
Hands-on research experience in bioinformatics, MRI, histopathology, or multiple sclerosis. Please note that clinical or diagnostic experience without a research component does not meet this criterion.

- Cardiovascular systems
- Human research
- Immunology
- Molecular and cell biology
Blood-contacting medical devices, such as heart pumps, prosthetic valves, and extracorporeal systems, expose blood to unnatural mechanical forces that can trigger both clotting and bleeding, limiting patient safety. This PhD project aims to understand how platelets, critical blood cells for clotting, respond to these mechanical stresses and to identify biomarkers that predict blood damage in individual patients. We combine laboratory experiments, patient studies, and computational simulations to map how platelets activate, aggregate, shed key receptors, and release extracellular vesicles under defined flow conditions. We will explore mechanistic responses in vitro, including the effects of anticoagulants, antiplatelet drugs, sex differences, and inflammatory or metabolic preconditioning, reflecting clinical diversity. Using these data, we aim to develop predictive models to guide personalized device settings and safer designs. Clinical validation will involve prospective patient studies and analysis of explanted devices to link observed platelet dysfunction with local flow patterns and thrombus formation. The outcomes are expected to define a panel of shear-sensitive platelet biomarkers, improve mechanistic understanding of device-related blood damage, and inform the design and clinical use of circulatory support systems. By combining mechanistic, computational, and translational approaches, this work addresses a major clinical challenge and has the potential to significantly improve patient safety and individualized cardiovascular care.
Skills to be acquired during PhD training
During the PhD project, the student will gain interdisciplinary training in blood cell biology, cardiovascular device research, and translational science, including:
- Advanced understanding of blood cell biology and the impact of shear stress on hemocompatibility
- Hands-on experience with flow-based mechanobiology assays and advanced imaging approaches
- Analysis of platelet function and extracellular vesicles in cardiovascular systems
- Exposure to translational and patient-centered research related to cardiovascular medical devices
- Training in computational modeling and the integration of experimental and clinical data
- Development of scientific communication and presentation skills, as well as experience in interdisciplinary collaboration
- Acquisition of transferable skills in project management, teamwork, and problem-solving across basic and translational research
PhD network and research environment
The project is embedded in the research environment of the Ludwig Boltzmann Institute for Cardiovascular Research, a transdisciplinary platform dedicated to innovative cardiovascular research.
The PhD student will benefit from:
- Access to the institute’s advanced research infrastructure and collaborative environment
- Interaction with an interdisciplinary team of scientists and clinicians
- Training opportunities and support services provided by the Ludwig Boltzmann Society
- Opportunities to engage in collaborative and translational research projects across disciplines
Master in Natural or Medical Sciences
Advantage: Experience with mouse models
- Behavioural biology
- Computer lab
- Data Science
- Human research
- Neuroscience
- Wet lab
Success in life often depends on inspired choices between risk-taking for exciting achievements or perseverance with safe, incremental advancements. In this project we aim to discover how GABAergic interneurons and pyramidal cells of the prefrontal cortex cooperate in time to generate such inspired decisions based on internal valuations of past experiences. We will train mice to perform a gambling-task, successfully adjusting choices between certain-small, or possible-big rewards with changing long-term advantages. Changes of choice strategy are reflected by abrupt changes of the neuronal firing rate in prefrontal networks. We aim to discover the neuronal mechanisms underlying and initiating these change points. We will perform juxtacellular recording and labelling of distinct types of GABAergic interneurons during the value-based decision making task to determine their cellular identity and axonal projections. This will be supplemented with large-scale extracellular Neuropixels recordings, Ca2+ imaging and optotagging of multiple interneurons and pyramidal cells simultaneously. We will determine the precise firing patterns of the interneurons in relation to the population activity and in relation to the choice behavior and switches of strategy following reward experiences. We will test if the variability, vicariousness and motivational state during choice selection can be explained by the firing of interneurons. Furthermore, we will detect network oscillations in the circuits of the prefrontal cortex and test their relation to decision making, behavioural strategy and spike timing of neurons. In this project will define the contribution of GABAergic interneurons to concerted network activity and decision making based on internal valuation of likely outcomes.
Skills to be acquired during PhD training
During the PhD project, the student will gain interdisciplinary training in systems neuroscience, neural circuit analysis, and behavioral neuroscience, including:
- In vivo electrophysiological recordings using glass electrodes and Neuropixels probes in freely moving and head-fixed rodents
- Application of optogenetic techniques to identify and manipulate specific neuronal populations
- Design and implementation of behavioral decision-making tasks in mice
- Calcium imaging to monitor neuronal population activity during behavior
- Analysis of multi-channel spike data and local field potentials
- Investigation of network oscillations and neuronal population dynamics during decision-making
- Immunofluorescence microscopy and histological analysis of neuronal identity
- Reconstruction of neuronal morphology using Neurolucida 3D neuron tracing
- Advanced computational analysis of neural and behavioral datasets
PhD network and research environment
The project will be conducted at the Medical University of Vienna within the Division of Cognitive Neurobiology.
The PhD student will also be part of the international research collaboration DFG Research Unit 5159 – Resolving Prefrontal Flexibility, funded by the German Research Foundation.
The research environment provides:
- A collaborative and interdisciplinary neuroscience environment
- Access to state-of-the-art electrophysiology, imaging, and behavioral neuroscience infrastructure
- Regular scientific meetings and workshops with international partner laboratories
- Opportunities for working visits and collaborative projects with partner labs in Germany
- Participation in a vibrant scientific community studying prefrontal circuit function and cognitive flexibility
Master degree in life science, computer science, natural science or medicine and should have a strong scientific background either in experimental investigations of neuronal circuits OR in computational data analysis
- Animal research
- Cardiovascular systems
- Human research
- Immunology
- NET‑IT DocFund
Histamine plays a crucial role in inflammation, immune response, and vasospasms of atherosclerotic coronary arteries. Interestingly, histamine is released after myocardial infarction, and this could contribute to arrhythmias. Histamine may also promote thrombosis through several mechanisms, including platelet aggregation and endothelial dysfunction, and may contribute to disseminated intravascular coagulation in anaphylaxis. Patients with anaphylaxis who develop thrombosis or bleeding have a dismal outcome, and many patients with acute asthma exacerbations develop pulmonary embolism, the mechanism of which is unclear. This thesis will explore potential mechanism by which histamine/anaphylaxis/asthma can induce thrombosis. The project integrates patient-oriented clinical research with applied translational laboratory approaches in animals. The PhD candidate will be actively involved in patient recruitment, interaction, and clinical data collection within structured clinical trial settings. Further the PhD candidate will learn to conduct animal trials being assisted by an experienced veterinarian. This interdisciplinary framework provides a unique opportunity to directly link clinical outcomes with mechanistic insights. The candidate will gain hands-on experience in the conduct of clinical trials, animal trials, data analysis, and modern laboratory techniques in immunology and vascular biology, while developing a strong translational research perspective. The project is embedded within the Department of Clinical Pharmacology at the Medical University of Vienna, offering access to well-established clinical research infrastructure, state-of-the-art core facilities, and a highly collaborative environment.
Skills to be acquired during PhD training
During this PhD project, the candidate will gain comprehensive, hands-on training at the interface of clinical research and applied biomedical science. The structured combination of patient-oriented work and animal experiments offers a unique opportunity to develop a broad and highly relevant skill set. The candidate will be actively involved in the conduct of randomized, double-blind, placebo-controlled clinical trials, including patient recruitment, informed consent procedures, and ICH compliant documentation.
A strong emphasis is placed on data handling, analysis, and interpretation. The candidate will learn how to integrate clinical endpoints with laboratory-derived data, enabling a deeper understanding of disease mechanisms and therapeutic effects. In addition, the candidate will have the opportunity to contribute to other ongoing projects within the department, allowing them to broaden their expertise across different research areas and methodologies. This exposure will further strengthen their scientific versatility and interdisciplinary perspective.
Importantly, the project fosters the development of a translational research mindset, teaching the candidate how to connect patient-centered observations with mechanistic insights. This combination of clinical and analytical expertise represents a highly valuable skill set for future careers in academia, clinical research, or the pharmaceutical and biotechnology sectors.
PhD network and research environment
The project is part of the FWF DocFund NET-IT (Dissecting the network of vascular inflammation and thrombosis). As a NET-IT PhD student, the candidate will join a doctoral college bringing together 10 research groups and a cohort of PhD students working on complementary aspects of vascular inflammation, thrombosis, and infection. The program provides an interdisciplinary setting that encourages scientific exchange and collaboration across laboratories.
The network provides:
- Internal and international laboratory rotations within partner research groups
- Structured training modules with yearly thematic focuses
- Courses on research methods, soft skills, grant writing, and scientific publishing
- Opportunities for interaction and collaboration with researchers from academia and industry
Required:
• Outstanding scholary performance
• MD degree at the time of project start
• Proficiency in German and English
• Documented interest in Clinical Pharmacology & Pulmonology
• Experience in writing scientific manuscripts
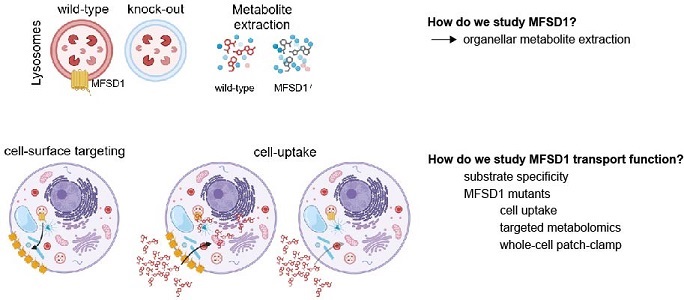
- Biochemistry
- Molecular and cell biology
- Wet lab
The proper distribution of small molecules (i.e., metabolites) between cell organelles and the cytoplasm, as well as between the cell’s interior and its exterior are of fundamental importance for normal functioning of cells, tissues, and organisms. Major guardians of the proper distribution of metabolites across cell membranes are membrane transporters of the solute carrier (SLC) protein family, which serves as selective gates. Malfunction of SLCs introduces a disbalance of metabolite distribution which frequently leads to a disease phenotype. Consequently, solute carriers are considered promising drug targets. Thus, the detailed description of the molecular function of novel SLCs will enhance the general understanding of their function and guide future approaches in drug development.
Recently, we have identified the lysosomal membrane protein MFSD1 to represent a new class of dipeptide transporters. MFSD1 is responsible for the lysosomal export of dipeptides, terminal breakdown products of lysosomal protein degradation. However, the exact mechanism by which MFSD1 selects its substrates, and how substrate binding initiates conformational change required for MFSD1 transport function is not understood.
Furthermore, we aim to understand the contribution of MFSD1 to normal cellular functioning. Thus, we will identify interaction partners of MFSD1 and study their contribution to lysosomal functioning. Additionally, we will investigate the lysosomal proteome and how it is affected by the absence of MFSD1. Our results will reveal MFSD1-dependent regulators of lysosomal biology and disclose its contribution to specific biological function.
Skills to be acquired during PhD training
During the PhD project, the student will gain comprehensive training in molecular and cellular biology, membrane transporter research, and functional analysis of lysosomal proteins, including:
- Generation of genetically modified cell lines using CRISPR–Cas9 genome editing
- Stable cell line generation using lentiviral vector systems
- Application of modern molecular cloning strategies and DNA manipulation techniques
- Analysis of protein expression and localization using confocal microscopy, Western blotting, and flow cytometry
- Quantitative gene expression analysis using PCR-based techniques (e.g., qPCR)
- Cell culture and functional characterization of membrane transporter proteins
- Application of omics approaches, including proteomics, metabolomics, and lipidomics
- Interpretation and integration of molecular and proteomic datasets to study lysosomal biology
- Development of scientific communication skills through presentations at seminars and international conferences
PhD network and research environment
The project will be conducted at the Medical University of Vienna in a research environment with strong expertise in solute carrier biology and membrane transporter research.
The PhD student will benefit from:
- A structured doctoral training program including seminars, workshops, and specialized courses in modern biomedical research methods
- Access to state-of-the-art infrastructure and molecular biology facilities
- Interaction with several experts in the field of solute carrier transporters
- Opportunities for scientific exchange and collaboration within the institute
- Participation in international conferences and internal research seminars
Masters degree in Molecular Biology (or in an equivalent field).
Proficiency in several molecular biological techniques (e.g., flow-cytometry, confocal microscopy, PCR, DNA cloning, qPCR, Western Blotting, cell culture, protein production/purification etc.) is expected.
Advantage: Knowledge in structural biology
- Animal research
- Cardiovascular systems
- Immunology
- Molecular and cell biology
- Neuroscience
- Wet lab
Hematopoiesis is a highly regulated stepwise process of blood cell differentiation from hematopoietic stem cells that takes place in the marrow of major bones. The link between hematopoiesis and hypercholesterolemia is widely documented in atherosclerosis that is the cause of myocardial infarction and many strokes. However, evidence is mainly based on studying long bones, and it remains unknown whether different bone compartments at various anatomical locations have divergent response to dyslipidemia. This project will investigate whether hypercholesterolemia affects hematopoiesis in the central nervous system (CNS), more precisely in the skull bone marrow, by studying the composition and function of its microenvironment as compared to long bones, and the distribution of skull-derived immune cells along various brain-border regions. Further, using state-of-the-art methodologies, we will study the contribution of the skull bone marrow to the circulating leukocyte pool and investigate its impact on atherosclerotic cardiovascular diseases. In the longer term, we will aim to understand how lifestyle interventions, such as sleep fragmentation, modify these processes. Our findings may identify candidate molecules as potential targets for instructing adequate hematopoiesis in a site-specific manner with critical relevance for multiple diseases concerning both the CNS (such as stroke and vascular dementia) as well as the periphery.
Skills to be acquired during PhD training
- Design and execution of advanced preclinical in vivo studies using state-of-the-art models of hematopoiesis and atherosclerosis
- Application of a wide-array of wet-lab techniques, including flow cytometry, histology, confocal microscopy as well as in vitro functional assays to investigate the proliferative capacity of hematopoietic stem cells
- Advanced molecular and histological techniques for tissue characterization of immunologically relevant brain-border regions, such as skull bone marrow, meninges, choroid plexus and draining cervical lymph nodes
- Application and integration of high-throughput omics approaches, such as RNA-sequencing and proteomics
- Development of scientific communication and writing, acquisition of skills related to data interpretation and critical reading of scientific literature, exposure to interdisciplinary research and international collaborations
Master’s degree in Biology, Molecular Biology, Biotechnology, or a related subject.
Advantage: Experience in working with mouse models
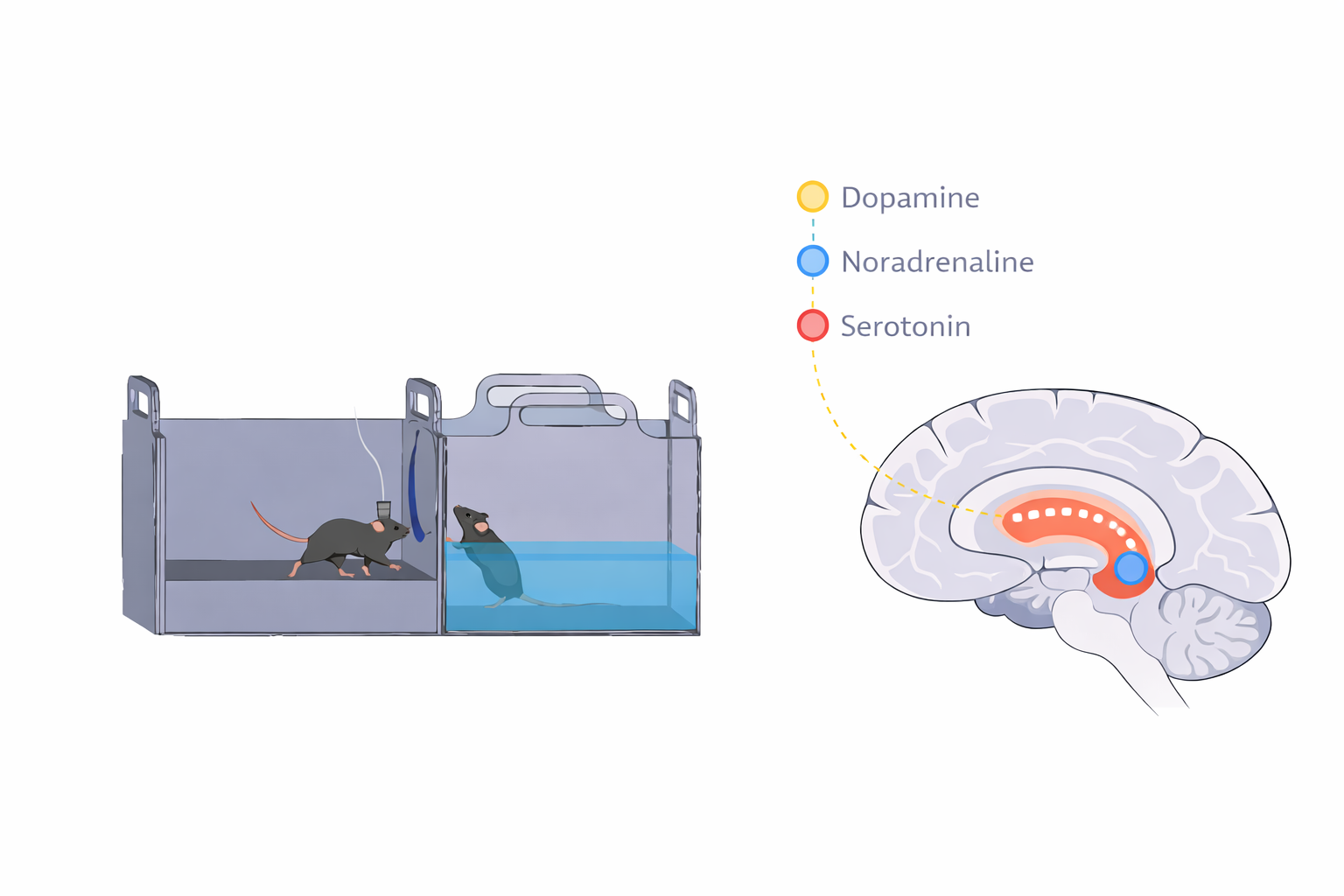
- Behavioural biology
- Data Science
- Molecular and cell biology
- Neuroscience
- Wet lab
This PhD position is part of the ERC-funded project “ProSocial: Regulation of Prosocial and Cognitive Behaviors along the Dorsoventral Hippocampal Axis.” The project aims to uncover the neural and cognitive mechanisms underlying helping behavior in mice, with the long-term goal of translating these findings to humans. Recent work from the Mikulovic Lab identified a distinct population of hippocampal neurons—termed “liberation cells”—that become active when a mouse frees another individual in distress (Dos Santos Correa et al., Cell Reports, 2026). Building on this discovery, the project will investigate the molecular identity of these cells and examine how neuromodulators such as dopamine, noradrenaline, and serotonin regulate prosocial behavior. A key objective is to understand how these neuromodulatory systems influence prosocial responses both in healthy animals and in disease models where prosociality is impaired. The PhD candidate will work with mouse models and should therefore have an interest in behavioral neuroscience; prior experience with animal models is advantageous. During the training, the candidate will gain hands-on experience with state-of-the-art techniques, including calcium and novel sensor imaging, optogenetics, and electrophysiology. Interest in advanced computational analysis of neural and behavioral data is also beneficial. As part of a recently funded ERC project, the successful candidate will join an interdisciplinary research environment dedicated to understanding the neural mechanisms of prosocial behavior across molecular, circuit, and behavioral levels.
Skills to be acquired during PhD training
During the PhD project, the student will gain interdisciplinary training in behavioral neuroscience, neural circuit analysis, and advanced neurotechnology, including:
- Design and execution of behavioral experiments in mouse models to study prosocial and cognitive behaviors
- Identification and characterization of specific neuronal populations involved in social behavior
- Application of calcium imaging and novel neural activity sensors to monitor neuronal dynamics in vivo
- Use of optogenetic approaches to manipulate neural circuits and investigate causal relationships in behavior
- Application of electrophysiological techniques to study neuronal activity and synaptic function
- Analysis of large-scale neural and behavioral datasets using advanced computational approaches
- Integration of molecular, circuit-level, and behavioral data to understand mechanisms of prosocial behavior
- Development of scientific communication, experimental design, and interdisciplinary collaboration skills
PhD network and research environment
The project is funded by the European Research Council (ERC) and will be conducted in an interdisciplinary neuroscience research environment at the Medical University of Vienna.
The PhD student will benefit from:
- A dynamic research environment focused on neural circuit mechanisms of social behavior
- Access to state-of-the-art neuroscience technologies for imaging, optogenetics, and electrophysiology
- Close collaboration with researchers working at the interface of neuroscience, behavior, and computational analysis
- Opportunities for scientific exchange through seminars, journal clubs, and international collaborations
- Participation in conferences and training activities supporting scientific and career development
Master’s background in Neuroscience, Biology, Biotechnology, Biomedical Engineering, or similar.
Skilled in animal behavioral paradigms, molecular biology techniques, and computational data analysis. Interested in bridging experimental neuroscience with data-driven approaches to investigate neural mechanisms underlying behavior and disease.
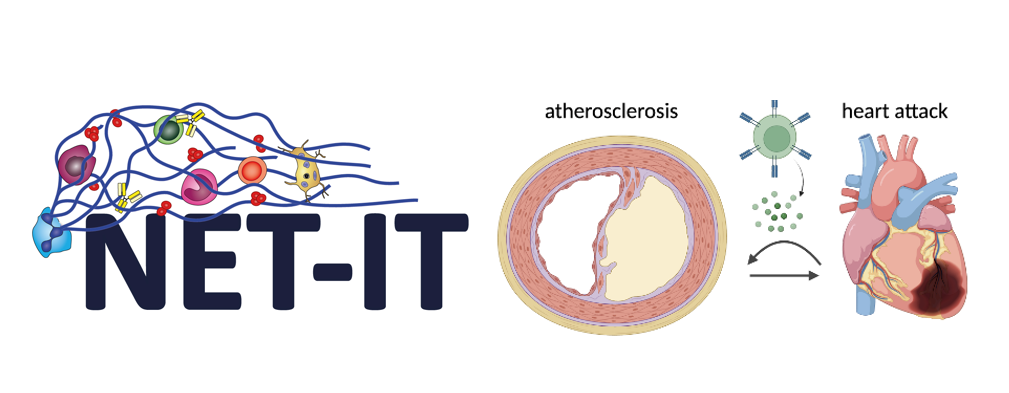
- Cardiovascular systems
- NET‑IT DocFund
- Wet lab
Patients who survive a heart attack (a condition known as myocardial infarction) are at particularly high risk of experiencing recurrent cardiovascular events. This increased risk is associated with an accelerated progression of atherosclerosis following the initial cardiac injury, which significantly contributes to long-term morbidity and mortality. However, the biological mechanisms that drive this post-myocardial infarction acceleration of atherosclerosis remain poorly understood. The immune system plays a central role in both the development of atherosclerosis and the repair processes following myocardial infarction. Among immune cells, T lymphocytes are key regulators of inflammatory responses and have been shown to influence plaque development, plaque stability, and cardiac remodelling after myocardial infarction. Different T cell subsets can exert both protective and detrimental effects depending on their activation state and functional properties. Despite their recognized importance, the role of T cells in driving the acceleration of atherosclerosis after myocardial infarction remains unexplored. This PhD project aims to investigate how myocardial infarction alters T cell responses and how these immune mechanisms contribute to the progression of atherosclerosis. By combining advanced preclinical models, immunological analyses, and high-throughput technologies, the project seeks to uncover novel mechanisms linking cardiac injury to systemic immune responses and atherosclerosis progression. A better understanding of these processes may reveal new therapeutic targets to prevent recurrent cardiovascular events and improve long-term outcomes for patients suffering from heart attacks.
Skills and techniques the PhD student will acquire during the project:
- Design and execution of advanced preclinical in vivo studies
- Expertise in wet-lab techniques: including flow cytometry, histology, confocal microscopy, and primary cell culture
- Application of high-throughput omics technologies, including RNA-sequencing and spatial transcriptomics
- Scientific communication and presentation of research findings
- Scientific writing and preparation of manuscripts and research reports
PhD program and research environment:
The project is embedded within the NET IT DocFund Program, providing access to a strong interdisciplinary training environment and a broad international network of collaborators. Through this network, the doctoral candidate will benefit from access to advanced technologies, shared expertise, and cutting-edge research tools. The work will be conducted in a vibrant, enthusiastic, and international research environment within the Tsiantoulas Group at the Medical University of Vienna, fostering collaboration, scientific exchange, and professional development.
Master’s degree in Biology, Molecular Biology, Biotechnology, or a related subject.
Advantage: Experience working with mouse models
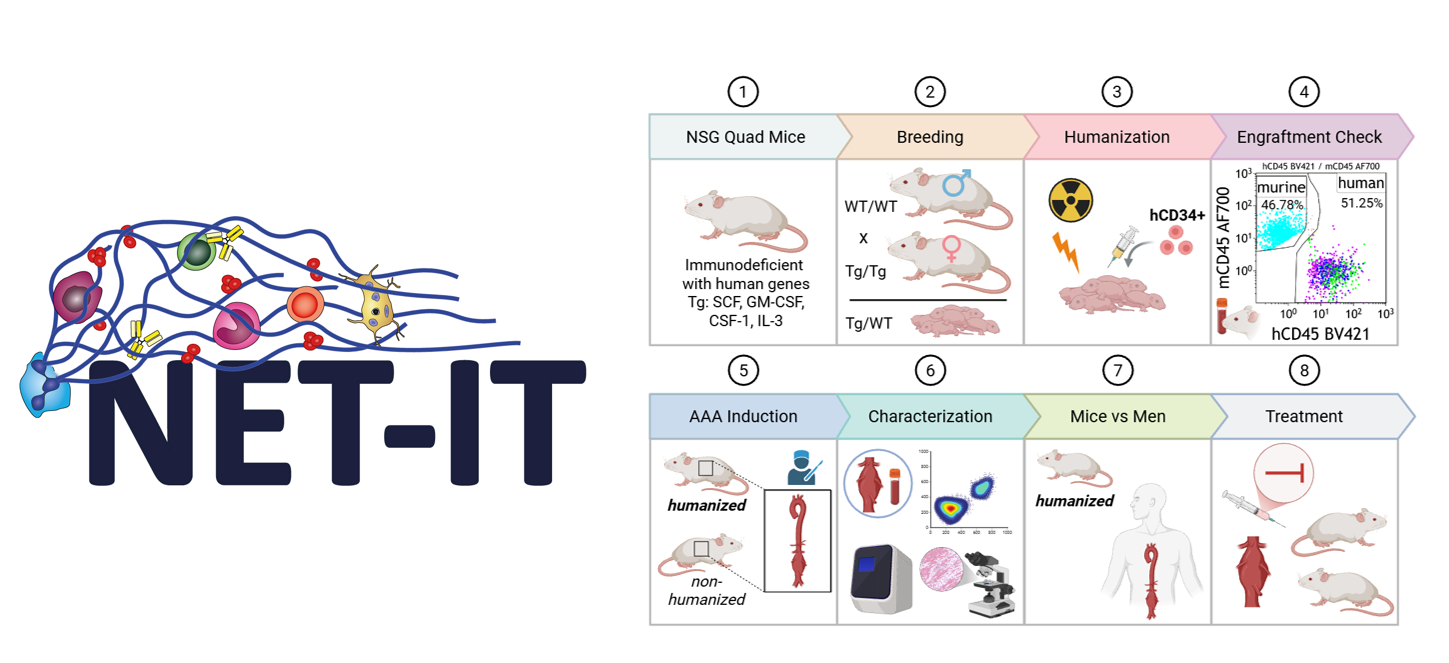
- Animal research
- Cardiovascular systems
- Drug targets and/or drug development
- Human research
- Immunology
- NET‑IT DocFund
- Wet lab
The pathological vessel dilatation of abdominal aortic aneurysms (AAAs) is a result of media destruction driven by pro-inflammatory and proteolytic activity in the vessel wall and the frequently occurring intraluminal thrombus. Available treatment to prevent the life-threatening event of aneurysm rupture is currently limited to high-risk surgery, since drug options to control aneurysm expansion are lacking. Therapeutics successful in AAA mouse models generally failed in clinical trials which has raised concerns regarding the aptness of the experimental models. Based on previous data we propose that the central interplay of leukocytes (inflammation) and platelets (thrombosis) may not be adequately represented in the experimental AAA models, since blood cell composition differs considerably between mice and men. As recommended by the FDA for preclinical drug testing, we intend to apply so-called “humanized mice” (with functional human myeloid, lymphoid and thrombocyte compartments) in models of AAA induction and treatment, to improve the reliable prediction of clinical success. In the proposed PhD project, we aim to compare resulting aneurysms in humanized versus non-humanized mice to human AAAs by a comprehensive histological, cellular and molecular characterization. Furthermore, the efficacy of thrombo-inflammatory drug candidates will be investigated in the humanized versus non-humanized models.
Skills to be acquired during PhD training
During the PhD project, the student will gain extensive training in experimental vascular biology and translational research, including:
- Generation of Humanized mouse models through transfer of cord-blood derived human CD34⁺ hematopoietic stem cells into immunocompromised animals
- Induction of experimental aneurysms using elastase- and angiotensin II-based mouse models of Abdominal Aortic Aneurysm
- Monitoring aneurysm development and growth using 3D ultrasound imaging
- Histological characterization of human and mouse aneurysms through immunohistochemistry and immunofluorescence staining
- Analysis of media destruction, inflammatory infiltrates, and thrombus formation in vascular tissue
- Investigation of molecular mechanisms underlying thrombo-inflammatory processes in AAA pathogenesis
- Experimental evaluation of potential thrombo-inflammatory therapeutic strategies
PhD network and research enviroment
The project is part of the FWF DocFund NET-IT (Dissecting the network of vascular inflammation and thrombosis). As a NET-IT PhD student, the candidate will join a doctoral college bringing together 10 research groups and a cohort of PhD students working on complementary aspects of vascular inflammation, thrombosis, and infection. The program provides an interdisciplinary setting that encourages scientific exchange and collaboration across laboratories.
The network provides:
- Internal and international laboratory rotations within partner research groups
- Structured training modules with yearly thematic focuses
- Courses on research methods, soft skills, grant writing, and scientific publishing
- Opportunities for interaction and collaboration with researchers from academia and industry
Master’s degree in natural or medical sciences; prior experience with mouse models (e.g., FELASA B–equivalent training) is preferred, and experience with humanized mouse models is considered an advantage.
- Biochemistry
- Drug targets and/or drug development
- Immunology
- Molecular and cell biology
- Wet lab
Systemic sclerosis (SSc) is a chronic disease marked by progressive fibrosis of the skin and internal organs. Despite recent advances, SSc continues to have a very poor prognosis. Thus, there is a substantial unmet medical need to identify novel drug targets. By mining publicly available next generation sequencing (NGS) datasets from SSc skin biopsy studies we identified NNMT as a potential drug target to overcome dermal fibroblast (DF) activation and fibrosis in SSc.
Research questions and objectives: This project is designed to prove the hypothesis that NNMT, a methyltransferase at the interface of metabolism and epigenetics, is a key regulator of DF activation and ECM overproduction. By gaining a deeper understanding of the molecular mechanisms underlying NNMT-driven activation of SSc DF we aim to pave the way for novel fibroblast-targeting therapies.
Approach and methods: We will combine advanced high-throughput omics technologies, cutting-edge molecular biology and computational tools to address the research questions of this project. Imaging mass cytometry of skin punch biopsies will determine the degree and distribution of NNMT expression in SSc and healthy skin. To dissect NNMT´s role in DF functions we will generate NNMT knockout (KO) and NNMT overexpressing DF cell lines using CRISPR/Cas9 gene editing. Functions and output of genetically edited DFs will be analysed using cell contraction assays and collagen detection ELISA. A high-content screening, image-based cell-cell profiling platform will be employed to investigate NNMT´s impact on pathogenic DF-immune cell interactions. High-throughput profiling of metabolites, mRNA expression, DNA methylation and histone modifications of genetically edited DFs will identify mechanisms and pathways that link NNMT activity with DF activation. Results will be integrated with previously published scRNA-seq data of the skin. Multimodal integration will facilitate the identification of NNMT-regulated cell-specific pathways important at several levels of biological organization. The in vivo relevance of NNMT in fibrotic disease will be assessed using a fibroblast-specific NNMT KO mouse model of dermal fibrosis.
Level of originality and innovation: There is an urgent unmet medical need to identify novel drug targets for SSc therapy. Although our preliminary results strongly point towards NNMT as a master regulator of DF activation and ECM overproduction in SSc this still needs to be proven. The strength of this project lies in the strategic and disease-specific integration of established techniques, rather than in the development of new methods. By applying these tools to an entirely unexplored axis of SSc pathogenesis, the project offers both conceptual and translational innovation.
Skills to be acquired during PhD training
- Gene editing strategies (CRISPR/Cas9 in primary cells)
- In vivo fibrosis models (fibroblast-specific KO mice)
- Multi-omics profiling (metabolomics, transcriptomics, epigenomics), including basic data analysis and interpretation
- Imaging Mass Cytometry
Functional fibroblast assays (e.g. collagen contraction assay, wounding healing assay)
PhD network and research environment
The PhD project is embedded in the Division of Rheumatology, providing a highly translational research environment that bridges basic science and clinical application. The candidate will work closely with clinicians and researchers, gaining insight into patient-oriented research in systemic sclerosis. The program includes participation in national and international conferences, as well as regular seminars and scientific exchanges within the division. This setting offers excellent opportunities for networking, interdisciplinary collaboration, and professional development.
Master’s degree in Biology, Molecular Biology, Biotechnology, or a related subject
- Familiarity with cell culture and basic molecular techniques
- Strong Interest in translational biomedical research
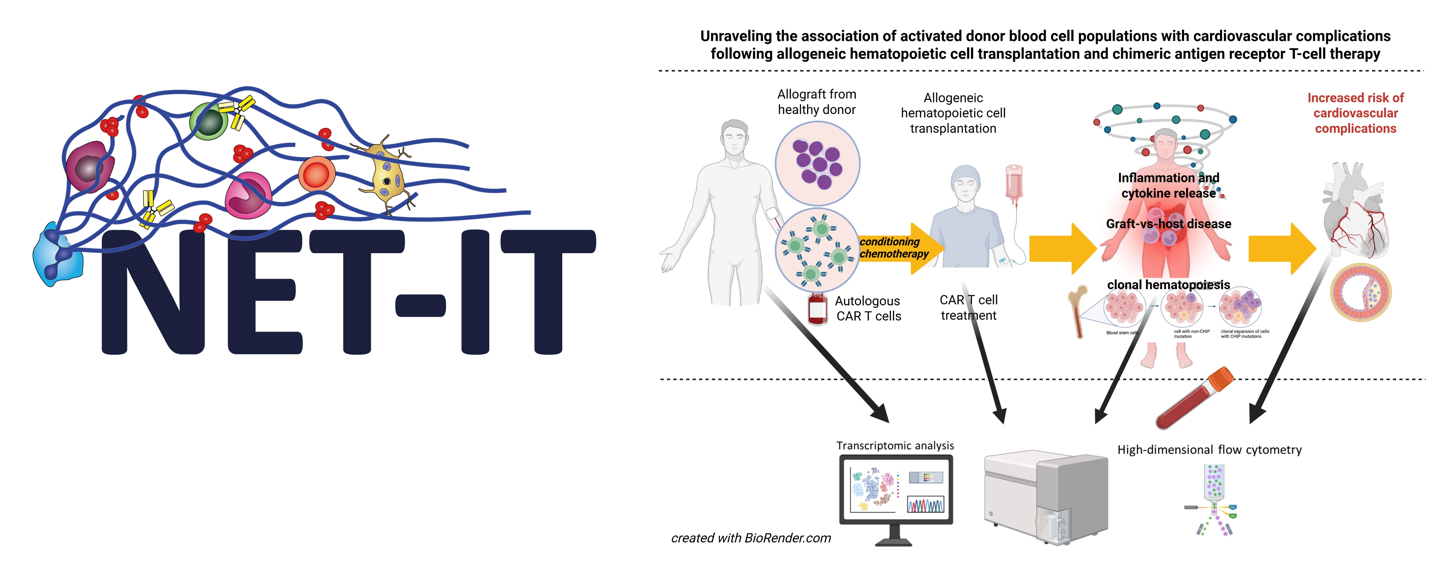
- Cancer
- Cardiovascular systems
- Immunology
- Molecular and cell biology
- NET‑IT DocFund
- Wet lab
Cardiovascular disease (CVD) is increasingly recognized as a major long-term complication in patients undergoing allogeneic hematopoietic cell transplantation (allo-HCT) and chimeric antigen receptor T-cell (CAR-T) therapy. Despite improved survival, the mechanisms driving post-treatment vascular complications remain poorly understood. Emerging evidence suggests that interactions between hematopoiesis and inflammation—highlighted by the link between clonal hematopoiesis and atherosclerosis—may play a central role. However, the contribution of donor-derived immune and blood cell populations to cardiovascular risk after these therapies has not yet been systematically investigated. This project aims to unravel the role of activated donor blood cell populations in the development of cardiovascular complications following allo-HCT and CAR-T therapy. We hypothesize that treatment-related vascular damage, combined with chronic immune activation, promotes a pro-atherogenic environment. In particular, activated platelets, monocytes/macrophages, and T cells may contribute to endothelial dysfunction and thrombotic plaque formation. To address this, we will comprehensively characterize immune cell activation profiles in patients with and without CVD after allo-HCT and CAR-T therapy. Using advanced techniques including multi-color flow cytometry, transcriptomic analysis, and serum biomarker profiling, we aim to identify cellular and molecular signatures associated with increased cardiovascular risk. Comparative analyses with control groups will further clarify whether these alterations are therapy-induced or pre-existing. Ultimately, this study seeks to identify novel biomarkers and mechanistic pathways underlying CVD in this vulnerable population, paving the way for improved risk stratification and targeted preventive strategies.
Skills to be acquired during PhD training
- immunological assays (cytotoxicity and cytokine secretion assays, phenotypic analysis by multicolour flow panels, signaling analysis by phosphoFlow)
- immunophenotyping
- cell transfection and transduction (lentiviral transduction or transient transfection)
- molecular biology (cloning, sequencing)
PhD network and research environment
The project is part of the FWF DocFund NET-IT (Dissecting the network of vascular inflammation and thrombosis). As a NET-IT PhD student, the candidate will join a doctoral college bringing together 10 research groups and a cohort of PhD students working on complementary aspects of vascular inflammation, thrombosis, and infection. The program provides an interdisciplinary setting that encourages scientific exchange and collaboration across laboratories.
The network provides:
- Internal and international laboratory rotations within partner research groups
- Structured training modules with yearly thematic focuses
- Courses on research methods, soft skills, grant writing, and scientific publishing
- Opportunities for interaction and collaboration with researchers from academia and industry
Experience with cell culture, immunophenotyping, molecular biology methods
- Drug targets and/or drug development
- Human research
- Neuroscience
The aim of this FWF-funded project is to assess whether modulation of peripheral ion channels can reduce pain. The project integrates experimental model development with randomized, placebo-controlled clinical trials to evaluate locally applied therapeutic approaches.
The PhD position will be Co-supervised by Prof. Dr. Stefan Heber
Skills to be Acquired During PhD Training
During the PhD, the candidate will develop competencies that are directly relevant for a career in clinical research and translational medicine, including:
- Planning, conduct, and statistical evaluation (biometry) of clinical trials
- Practical experience in randomized, placebo-controlled human studies
- Application of quantitative sensory testing and psychophysical methods in patients
- Interpretation of clinical data in the context of underlying biological mechanisms
- Use of molecular and functional assays to investigate therapeutic targets
- Integration of clinical observations with experimental findings
- Critical assessment of study design, data quality, and reproducibility
- Scientific writing and communication for clinical and translational research
These skills provide a foundation for future roles in academic medicine, clinical trials, or industry-driven drug development.
PhD network and research environment
The project is conducted within a clinical study at AKH and the Medical University of Vienna and is embedded in the structured PhD program in Molecular Signal Transduction.
The candidate will be directly involved in the execution of a clinical trial while contributing to the mechanistic understanding of therapeutic interventions. This setup allows medical graduates to extend their clinical training into research without losing clinical relevance.
The position provides experience that is typically not part of standard clinical training, particularly in trial design, data analysis, and the development of new therapeutic approaches. The combination of clinical study involvement and experimental work enables the candidate to build a profile at the interface of medicine and research.
Training is provided through supervised project work, local coursework, and integration into an interdisciplinary team across clinical and experimental settings.
Medical degree (completed or near completion) required for clinical study participation; other educational backgrounds cannot be considered.
Advantageous: Experience in a basic research laboratory